An official website of the United States government
Official websites use .gov A .gov website belongs to an official government organization in the United States.
Secure .gov websites use HTTPS A lock ( Lock Locked padlock icon ) or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.
- Publications
- Account settings
- Advanced Search
- Journal List


Dementia prevention, intervention, and care: 2020 report of the Lancet Commission
Gill livingston , prof, md, jonathan huntley , phd, andrew sommerlad , phd, david ames , prof, md, clive ballard , prof, md, sube banerjee , prof, md, carol brayne , prof, md, alistair burns , prof, md, jiska cohen-mansfield , prof, phd, claudia cooper , prof, phd, sergi g costafreda , phd, amit dias , md, nick fox , prof, md, laura n gitlin , prof, phd, robert howard , prof, md, helen c kales , prof, md, mika kivimäki , prof, fmedsci, eric b larson , prof, md, adesola ogunniyi , prof, mbchb, vasiliki orgeta , phd, karen ritchie , prof, phd, kenneth rockwood , prof, md, elizabeth l sampson , prof, md, quincy samus , phd, lon s schneider , prof, md, geir selbæk , prof, md, linda teri , prof, phd, naaheed mukadam , phd.
- Author information
- Article notes
- Copyright and License information
Correspondence to: Prof Gill Livingston, Division of Psychiatry, University College London, London W1T 7NF, UK
Issue date 2020 8-14 August.
Since January 2020 Elsevier has created a COVID-19 resource centre with free information in English and Mandarin on the novel coronavirus COVID-19. The COVID-19 resource centre is hosted on Elsevier Connect, the company's public news and information website. Elsevier hereby grants permission to make all its COVID-19-related research that is available on the COVID-19 resource centre - including this research content - immediately available in PubMed Central and other publicly funded repositories, such as the WHO COVID database with rights for unrestricted research re-use and analyses in any form or by any means with acknowledgement of the original source. These permissions are granted for free by Elsevier for as long as the COVID-19 resource centre remains active.
Executive summary
The number of older people, including those living with dementia, is rising, as younger age mortality declines. However, the age-specific incidence of dementia has fallen in many countries, probably because of improvements in education, nutrition, health care, and lifestyle changes. Overall, a growing body of evidence supports the nine potentially modifiable risk factors for dementia modelled by the 2017 Lancet Commission on dementia prevention, intervention, and care: less education, hypertension, hearing impairment, smoking, obesity, depression, physical inactivity, diabetes, and low social contact. We now add three more risk factors for dementia with newer, convincing evidence. These factors are excessive alcohol consumption, traumatic brain injury, and air pollution. We have completed new reviews and meta-analyses and incorporated these into an updated 12 risk factor life-course model of dementia prevention. Together the 12 modifiable risk factors account for around 40% of worldwide dementias, which consequently could theoretically be prevented or delayed. The potential for prevention is high and might be higher in low-income and middle-income countries (LMIC) where more dementias occur.
Our new life-course model and evidence synthesis has paramount worldwide policy implications. It is never too early and never too late in the life course for dementia prevention. Early-life (younger than 45 years) risks, such as less education, affect cognitive reserve; midlife (45–65 years), and later-life (older than 65 years) risk factors influence reserve and triggering of neuropathological developments. Culture, poverty, and inequality are key drivers of the need for change. Individuals who are most deprived need these changes the most and will derive the highest benefit.
Policy should prioritise childhood education for all. Public health initiatives minimising head injury and decreasing harmful alcohol drinking could potentially reduce young-onset and later-life dementia. Midlife systolic blood pressure control should aim for 130 mm Hg or lower to delay or prevent dementia. Stopping smoking, even in later life, ameliorates this risk. Passive smoking is a less considered modifiable risk factor for dementia. Many countries have restricted this exposure. Policy makers should expedite improvements in air quality, particularly in areas with high air pollution.
We recommend keeping cognitively, physically, and socially active in midlife and later life although little evidence exists for any single specific activity protecting against dementia. Using hearing aids appears to reduce the excess risk from hearing loss. Sustained exercise in midlife, and possibly later life, protects from dementia, perhaps through decreasing obesity, diabetes, and cardiovascular risk. Depression might be a risk for dementia, but in later life dementia might cause depression. Although behaviour change is difficult and some associations might not be purely causal, individuals have a huge potential to reduce their dementia risk.
In LMIC, not everyone has access to secondary education; high rates of hypertension, obesity, and hearing loss exist, and the prevalence of diabetes and smoking are growing, thus an even greater proportion of dementia is potentially preventable.
Amyloid-β and tau biomarkers indicate risk of progression to Alzheimer's dementia but most people with normal cognition with only these biomarkers never develop the disease. Although accurate diagnosis is important for patients who have impairments and functional concerns and their families, no evidence exists to support pre-symptomatic diagnosis in everyday practice.
Our understanding of dementia aetiology is shifting, with latest description of new pathological causes. In the oldest adults (older than 90 years), in particular, mixed dementia is more common. Blood biomarkers might hold promise for future diagnostic approaches and are more scalable than CSF and brain imaging markers.
Wellbeing is the goal of much of dementia care. People with dementia have complex problems and symptoms in many domains. Interventions should be individualised and consider the person as a whole, as well as their family carers. Evidence is accumulating for the effectiveness, at least in the short term, of psychosocial interventions tailored to the patient's needs, to manage neuropsychiatric symptoms. Evidence-based interventions for carers can reduce depressive and anxiety symptoms over years and be cost-effective.
Keeping people with dementia physically healthy is important for their cognition. People with dementia have more physical health problems than others of the same age but often receive less community health care and find it particularly difficult to access and organise care. People with dementia have more hospital admissions than other older people, including for illnesses that are potentially manageable at home. They have died disproportionately in the COVID-19 epidemic. Hospitalisations are distressing and are associated with poor outcomes and high costs. Health-care professionals should consider dementia in older people without known dementia who have frequent admissions or who develop delirium. Delirium is common in people with dementia and contributes to cognitive decline. In hospital, care including appropriate sensory stimulation, ensuring fluid intake, and avoiding infections might reduce delirium incidence.
Key messages.
Three new modifiable risk factors for dementia
- • New evidence supports adding three modifiable risk factors—excessive alcohol consumption, head injury, and air pollution—to our 2017 Lancet Commission on dementia prevention, intervention, and care life-course model of nine factors (less education, hypertension, hearing impairment, smoking, obesity, depression, physical inactivity, diabetes, and infrequent social contact).
Modifying 12 risk factors might prevent or delay up to 40% of dementias.
Be ambitious about prevention
- • Prevention is about policy and individuals. Contributions to the risk and mitigation of dementia begin early and continue throughout life, so it is never too early or too late. These actions require both public health programmes and individually tailored interventions. In addition to population strategies, policy should address high-risk groups to increase social, cognitive, and physical activity; and vascular health.
Specific actions for risk factors across the life course
- • Aim to maintain systolic BP of 130 mm Hg or less in midlife from around age 40 years (antihypertensive treatment for hypertension is the only known effective preventive medication for dementia).
- • Encourage use of hearing aids for hearing loss and reduce hearing loss by protection of ears from excessive noise exposure.
- • Reduce exposure to air pollution and second-hand tobacco smoke.
- • Prevent head injury.
- • Limit alcohol use, as alcohol misuse and drinking more than 21 units weekly increase the risk of dementia.
- • Avoid smoking uptake and support smoking cessation to stop smoking, as this reduces the risk of dementia even in later life.
- • Provide all children with primary and secondary education.
- • Reduce obesity and the linked condition of diabetes. Sustain midlife, and possibly later life physical activity.
- • Addressing other putative risk factors for dementia, like sleep, through lifestyle interventions, will improve general health.
Tackle inequality and protect people with dementia
- • Many risk factors cluster around inequalities, which occur particularly in Black, Asian, and minority ethnic groups and in vulnerable populations. Tackling these factors will involve not only health promotion but also societal action to improve the circumstances in which people live their lives. Examples include creating environments that have physical activity as a norm, reducing the population profile of blood pressure rising with age through better patterns of nutrition, and reducing potential excessive noise exposure.
- • Dementia is rising more in low-income and middle-income countries (LMIC) than in high-income countries, because of population ageing and higher frequency of potentially modifiable risk factors. Preventative interventions might yield the largest dementia reductions in LMIC.
For those with dementia, recommendations are:
Provide holistic post-diagnostic care
- • Post-diagnostic care for people with dementia should address physical and mental health, social care, and support. Most people with dementia have other illnesses and might struggle to look after their health and this might result in potentially preventable hospitalisations.
Manage neuropsychiatric symptoms
- • Specific multicomponent interventions decrease neuropsychiatric symptoms in people with dementia and are the treatments of choice. Psychotropic drugs are often ineffective and might have severe adverse effects.
Care for family carers
- • Specific interventions for family carers have long-lasting effects on depression and anxiety symptoms, increase quality of life, are cost-effective and might save money.
Acting now on dementia prevention, intervention, and care will vastly improve living and dying for individuals with dementia and their families, and thus society.
Introduction
Worldwide around 50 million people live with dementia, and this number is projected to increase to 152 million by 2050, 1 rising particularly in low-income and middle-income countries (LMIC) where around two-thirds of people with dementia live. 1 Dementia affects individuals, their families, and the economy, with global costs estimated at about US$1 trillion annually. 1
We reconvened the 2017 Lancet Commission on dementia prevention, intervention, and care 2 to identify the evidence for advances likely to have the greatest impact since our 2017 paper and build on its work. Our interdisciplinary, international group of experts presented, debated, and agreed on the best available evidence. We adopted a triangulation framework evaluating the consistency of evidence from different lines of research and used that as the basis to evaluate evidence. We have summarised best evidence using, where possible, good- quality systematic reviews, meta-analyses, or individual studies, where these add important knowledge to the field. We performed systematic literature reviews and meta-analyses where needed to generate new evidence for our analysis of potentially modifiable risk factors for dementia. Within this framework, we present a narrative synthesis of evidence including systematic reviews and meta-analyses and explain its balance, strengths, and limitations. We evaluated new evidence on dementia risk in LMIC; risks and protective factors for dementia; detection of Alzheimer's disease; multimorbidity in dementia; and interventions for people affected by dementia.
Nearly all the evidence is from studies in high-income countries (HIC), so risks might differ in other countries and interventions might require modification for different cultures and environments. This notion also underpins the critical need to understand the dementias related to life-course disadvantage—whether in HICs or LMICs.
Our understanding of dementia aetiology is shifting. A consensus group, for example, has described hippocampal sclerosis associated with TDP-43 proteinopathy, as limbic-predominant age-related TDP-43 encephalopathy (LATE) dementia, usually found in people older than 80 years, progressing more slowly than Alzheimer's disease, detectable at post-mortem, often mimicking or comorbid with Alzheimer's disease. 3 This situation reflects increasing attention as to how clinical syndromes are and are not related to particular underlying pathologies and how this might change across age. More work is needed, however, before LATE can be used as a valid clinical diagnosis.
The fastest growing demographic group in HIC is the oldest adults, those aged over 90 years. Thus a unique opportunity exists to focus on both human biology, in this previously rare population, as well as on meeting their needs and promoting their wellbeing.
Prevention of dementia
The number of people with dementia is rising. Predictions about future trends in dementia prevalence vary depending on the underlying assumptions and geographical region, but generally suggest substantial increases in overall prevalence related to an ageing population. For example, according to the Global Burden of Diseases, Injuries, and Risk Factors Study, the global age-standardised prevalence of dementia between 1990 and 2016 was relatively stable, but with an ageing and bigger population the number of people with dementia has more than doubled since 1990. 4
However, in many HIC such as the USA, the UK, and France, age-specific incidence rates are lower in more recent cohorts compared with cohorts from previous decades collected using similar methods and target populations 5 ( figure 1 ) and the age-specific incidence of dementia appears to decrease. 6 All-cause dementia incidence is lower in people born more recently, 7 probably due to educational, socio-economic, health care, and lifestyle changes. 2 , 5 However, in these countries increasing obesity and diabetes and declining physical activity might reverse this trajectory. 8 , 9 In contrast, age-specific dementia prevalence in Japan, South Korea, Hong Kong, and Taiwan looks as if it is increasing, as is Alzheimer's in LMIC, although whether diagnostic methods are always the same in comparison studies is unclear. 5 , 6 , 7
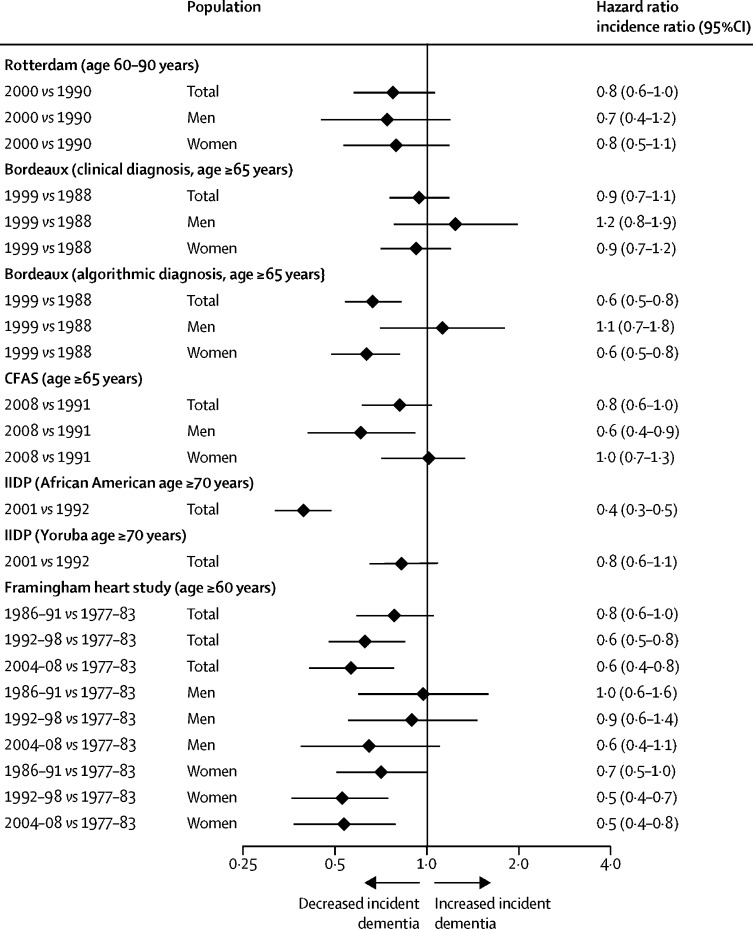
Incidence rate ratio comparing new cohorts to old cohorts from five studies of dementia incidence 5
IIDP Project in USA and Nigeria, Bordeaux study in France, and Rotterdam study in the Netherlands adjusted for age. Framingham Heart Study, USA, adjusted for age and sex. CFAS in the UK adjusted for age, sex, area, and deprivation. However, age-specific dementia prevalence is increasing in some other countries. IID=Indianapolis–Ibadan Dementia. CFAS=Cognitive Function and Ageing Study. Adapted from Wu et al, 5 by permission of Springer Nature.
Modelling of the UK change suggests a 57% increase in the number of people with dementia from 2016 to 2040, 70% of that expected if age-specific incidence rates remained steady, 10 such that by 2040 there will be 1·2 million UK people with dementia. Models also suggest that there will be future increases both in the number of individuals who are independent and those with complex care needs. 6
In our first report, the 2017 Commission described a life-course model for potentially modifiable risks for dementia. 2 Life course is important when considering risk, for example, obesity and hypertension in midlife predict future dementia, but both weight and blood pressure usually fall in later life in those with or developing dementia, 9 so lower weight and blood pressure in later life might signify illness, not an absence of risk. 11 , 12 , 13 , 14 We consider evidence on other potential risk factors and incorporate those with good quality evidence in our model.
Figure 2 summarises possible mechanisms of protection from dementia, some of which involve increasing or maintaining cognitive reserve despite pathology and neuropathological damage. There are different terms describing the observed differential susceptibility to age-related and disease-related changes and these are not used consistently. 15 , 16 A consensus paper defines reserve as a concept accounting for the difference between an individual's clinical picture and their neuropathology. It, divides the concept further into neurobiological brain reserve (eg, numbers of neurones and synapses at a given timepoint), brain maintenance (as neurobiological capital at any timepoint, based on genetics or lifestyle reducing brain changes and pathology development over time) and cognitive reserve as adaptability enabling preservation of cognition or everyday functioning in spite of brain pathology. 15 Cognitive reserve is changeable and quantifying it uses proxy measures such as education, occupational complexity, leisure activity, residual approaches (the variance of cognition not explained by demographic variables and brain measures), or identification of functional networks that might underlie such reserve. 15 , 16 , 17 , 18 , 19 , 20
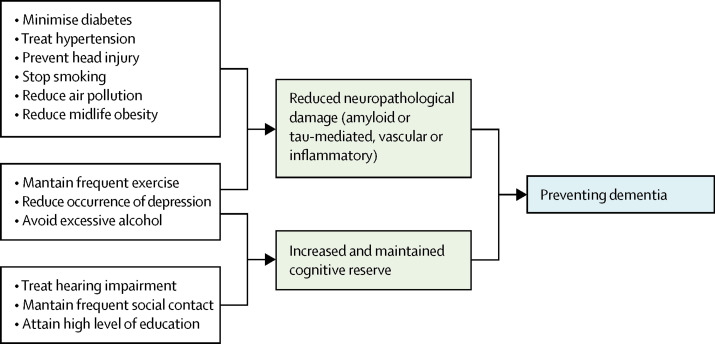
Possible brain mechanisms for enhancing or maintaining cognitive reserve and risk reduction of potentially modifiable risk factors in dementia
Early-life factors, such as less education, affect the resulting cognitive reserve. Midlife and old-age risk factors influence age-related cognitive decline and triggering of neuropathological developments. Consistent with the hypothesis of cognitive reserve is that older women are more likely to develop dementia than men of the same age, probably partly because on average older women have had less education than older men. Cognitive reserve mechanisms might include preserved metabolism or increased connectivity in temporal and frontal brain areas. 17 , 18 , 19 , 20 , 21 People in otherwise good physical health can sustain a higher burden of neuropathology without cognitive impairment. 22 Culture, poverty, and inequality are important obstacles to, and drivers of, the need for change to cognitive reserve. Those who are most deprived need these changes the most and will derive the highest benefit from them.
Smoking increases air particulate matter, and has vascular and toxic effects. 23 Similarly air pollution might act via vascular mechanisms. 24 Exercise might reduce weight and diabetes risk, improve cardiovascular function, decrease glutamine, or enhance hippocampal neurogenesis. 25 Higher HDL cholesterol might protect against vascular risk and inflammation accompanying amyloid-β (Aβ) pathology in mild cognitive impairment. 26
Dementia in LMIC
Numbers of people with dementia in LMIC are rising faster than in HIC because of increases in life expectancy and greater risk factor burden. We previously calculated that nine potentially modifiable risk factors together are associated with 35% of the population attributable fraction (PAFs) of dementia worldwide: less education, high blood pressure, obesity, hearing loss, depression, diabetes, physical inactivity, smoking, and social isolation, assuming causation. 2 Most research data for this calculation came from HIC and there is a relative absence of specific evidence of the impact of risk factors on dementia risk in LMIC, particularly from Africa and Latin America. 27
Calculations considering country-specific prevalence of the nine potentially modifiable risk factors indicate PAF of 40% in China, 41% in India and 56% in Latin America with the potential for these numbers to be even higher depending on which estimates of risk factor frequency are used. 28 , 29 Therefore a higher potential for dementia prevention exists in these countries than in global estimates that use data predominantly from HIC. If not currently in place, national policies addressing access to education, causes and management of high blood pressure, causes and treatment of hearing loss, socio-economic and commercial drivers of obesity, could be implemented to reduce risk in many countries. The higher social contact observed in the three LMIC regions provides potential insights for HIC on how to influence this risk factor for dementia. 30 We could not consider other risk factors such as poor health in pregnancy of malnourished mothers, difficult births, early life malnutrition, survival with heavy infection burdens alongside malaria and HIV, all of which might add to the risks in LMIC.
Diabetes is very common and cigarette smoking is rising in China while falling in most HIC. 31 A meta-analysis found variation of the rates of dementia within China, with a higher prevalence in the north and lower prevalence in central China, estimating 9·5 million people are living with dementia, whereas a slightly later synthesis estimated a higher prevalence of around 11 million. 30 , 32 These data highlight the need for more focused work in LMIC for more accurate estimates of risk and interventions tailored to each setting.
Specific potentially modifiable risk factors for dementia
Risk factors in early life (education), midlife (hypertension, obesity, hearing loss, traumatic brain injury, and alcohol misuse) and later life (smoking, depression, physical inactivity, social isolation, diabetes, and air pollution) can contribute to increased dementia risk ( table 1 ). Good evidence exists for all these risk factors although some late-life factors, such as depression, possibly have a bidirectional impact and are also part of the dementia prodrome. 33 , 34
PAF for 12 dementia risk factors
| Less education | 1·6 (1·3–2·0) | 40·0% | 61·2% | 19·4% | 7·1% |
| Hearing loss | 1·9 (1·4–2·7) | 31·7% | 45·6% | 22·2% | 8·2% |
| Traumatic brain injury | 1·8 (1·5–2·2) | 12·1% | 55·2% | 9·2% | 3·4% |
| Hypertension | 1·6 (1·2–2·2) | 8·9% | 68·3% | 5·1% | 1·9% |
| Alcohol (>21 units/week) | 1·2 (1·1–1·3) | 11·8% | 73·3% | 2·1% | 0·8% |
| Obesity (body-mass index ≥30) | 1·6 (1·3–1·9) | 3·4% | 58·5% | 2·0% | 0·7% |
| Smoking | 1·6 (1·2–2·2) | 27·4% | 62·3% | 14·1% | 5·2% |
| Depression | 1·9 (1·6–2·3) | 13·2% | 69·8% | 10·6% | 3·9% |
| Social isolation | 1·6 (1·3–1·9) | 11·0% | 28·1% | 4·2% | 3·5% |
| Physical inactivity | 1·4 (1·2–1·7) | 17·7% | 55·2% | 9·6% | 1·6% |
| Diabetes | 1·5 (1·3–1·8) | 6·4% | 71·4% | 3·1% | 1·1% |
| Air pollution | 1·1 (1·1–1·1) | 75·0% | 13·3% | 6·3% | 2·3% |
Data are relative risk (95% CI) or %. Overall weighted PAF=39·7%. PAF=population attributable fraction.
Weighted PAF is the relative contribution of each risk factor to the overall PAF when adjusted for communality.
In the next section, we briefly describe relevant newly published and illustrative research studies that add to the 2017 Commission's evidence base, including risks and, for some, mitigation. We have chosen studies that are large and representative of the populations, or smaller studies in areas where very little evidence exists. We discuss them in life-course order and within the life course in the order of magnitude of population attributable factor.
Education and midlife and late-life cognitive stimulation
Education level reached.
Higher childhood education levels and lifelong higher educational attainment reduce dementia risk. 2 , 35 , 36 , 37 New work suggests overall cognitive ability increases, with education, before reaching a plateau in late adolescence, when brain reaches greatest plasticity; with relatively few further gains with education after age 20 years. 38 This suggests cognitive stimulation is more important in early life; much of the apparent later effect might be due to people of higher cognitive function seeking out cognitively stimulating activities and education. 38 It is difficult to separate out the specific impact of education from the effect of overall cognitive ability, 38 , 39 and the specific impact of later-life cognitive activity from lifelong cognitive function and activity. 39 , 40
Cognitive maintenance
One large study in China tried to separate cognitive activity in adulthood from activities for those with more education, by considering activities judged to appeal to people of different levels of education. 40 It found people older than 65 years who read, played games, or bet more frequently had reduced risk of dementia (n=15 882, odds ratio [OR]=0·7, 95% CI 0·6–0·8). The study excluded people developing dementia less than 3 years after baseline to reduce reverse causation.
This finding is consistent with small studies of midlife activities which find them associated with better late-life cognition; so for example, in 205 people aged 30–64 years, followed up until 66–88 years, travel, social outings, playing music, art, physical activity, reading, and speaking a second language, were associated with maintaining cognition, independent of education, occupation, late-life activities, and current structural brain health. 41 Similarly, engaging in intellectual activity as adults, particularly problem solving, for 498 people born in 1936, was associated with cognitive ability acquisition, although not the speed of decline. 42
Cognitive decline
The use it or lose it hypothesis suggests that mental activity, in general, might improve cognitive function. People in more cognitively demanding jobs tend to show less cognitive deterioration before, and sometimes after retirement than those in less demanding jobs. 43 , 44 One systematic review of retirement and cognitive decline found conflicting evidence. 45 Subsequently, a 12-year study of 1658 people found older retirement age but not number of years working, was associated with lower dementia risk. 46 Those retiring because of ill health had lower verbal memory and fluency scores than those retiring for other reasons. 47 Another study found a two-fold increase in episodic memory loss attributable to retirement (n=18 575, mean age 66 years), compared to non-retirees, adjusting for health, age, sex, and wealth. 48 Similarly, in a cohort of 3433 people retiring at a mean age of 61 years, verbal memory declined 38% (95% CI 22–60) faster than before retirement. 44 In countries with younger compared to higher retirement ages, average cognitive performance drops more. 49
Cognitive interventions in normal cognition and mild cognitive impairment
A cognitive intervention or cognition-orientated treatment comprises strategies or skills to improve general or specific areas of cognition. 50 Computerised cognitive training programmes have increasingly replaced tasks that were originally paper-and-pencil format with computer-based tasks for practice and training. 51
Three systematic reviews in the general population found no evidence of generalised cognition improvement from specific cognitive interventions, including computerised cognitive training, although the domain trained might improve. 52 , 53 , 54
A meta-analysis of 17 controlled trials of at least 4 hours of computerised cognitive training, (n=351, control n=335) for mild cognitive impairment, found a moderate effect on general cognition post-training (Hedges' g=0·4, 0·2–0·5); 55 however few high quality studies and no long-term high quality evidence about prevention of dementia currently exists. A meta-analysis of 30 trials of computerised, therapy-based and multimodal interventions for mild cognitive impairment found an effect on activities of daily living (d=0·23) and metacognitive outcomes (d=0·30) compared to control. 56 A third systematic review identified five high quality studies, four group-delivered and one by computer, and concluded the evidence for the effects of cognitive training in mild cognitive impairment was insufficient to draw conclusions. 53 A comprehensive, high quality, systematic overview of meta-analyses of cognitive training in healthy older people, those with mild cognitive impairment and those with dementia, found that most were of low standard, were positive and most reached statistical significance but it was unclear whether results were of clinical value because of the poor standard of the studies and heterogeneity of results ( figure 3 ). 51
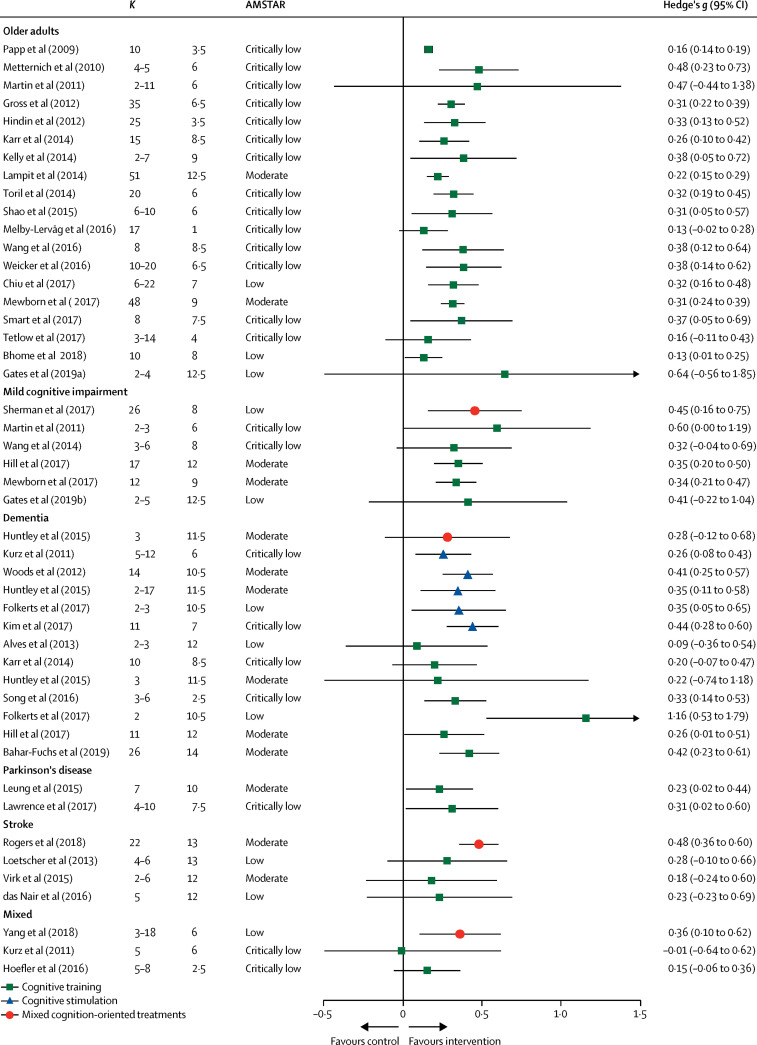
Pooled results of meta-analyses investigating objective cognitive outcomes of cognition-oriented treatment in older adults with and without cognitive impairment
K represents the number of primary trials included in the analysis. If a review reported several effect sizes within each outcome domain, a composite was created and k denotes the range of the number of primary trials that contributed to the effect estimate. AMSTAR=A MeaSurement Tool to Assess systematic Reviews (max score 16). Adapted from Gavelin et al, 51 by permission of Springer Nature.
In the only randomised controlled trial (RCT) of behavioural activation (221 people) for cognition in amnestic mild cognitive impairment, behavioural activation versus supportive therapy was associated with a decreased 2-year incidence of memory decline (relative risk [RR] 0·12, 0·02–0·74). 57
Hearing impairment
Hearing loss had the highest PAF for dementia in our first report, using a meta-analysis of studies of people with normal baseline cognition and hearing loss present at a threshold of 25 dB, which is the WHO threshold for hearing loss. In the 2017 Commission, we found an RR of 1·9 for dementia in populations followed up over 9–17 years, with the long follow-up times making reverse causation bias unlikely. 2 A subsequent meta-analysis using the same three prospective studies measuring hearing using audiometry at baseline, found an increased risk of dementia (OR 1·3, 95% CI 1·0–1·6) per 10 dB of worsening of hearing loss. 58 A cross-sectional study of 6451 individuals designed to be representative of the US population, with a mean age of 59·4 years, found a decrease in cognition with every 10 dB reduction in hearing, which continued to below the clinical threshold so that subclinical levels of hearing impairment (below 25 dB) were significantly related to lower cognition. 59
Although the aetiology still needs further clarification, a small US prospective cohort study of 194 adults without baseline cognitive impairment, (baseline mean age 54·5 years), and at least two brain MRIs, with a mean of 19 years follow-up, found that midlife hearing impairment measured by audiometry, is associated with steeper temporal lobe volume loss, including in the hippocampus and entorhinal cortex. 60
Hearing aids
A 25-year prospective study of 3777 people aged 65 years or older found increased dementia incidence in those with self-reported hearing problems except in those using hearing aids. 61 Similarly, a cross–sectional study found hearing loss was only associated with worse cognition in those not using hearing aids. 62 A US nationally representative survey of 2040 people older than 50 years, tested every two years for 18 years, found immediate and delayed recall deteriorated less after initiation of hearing aid use, adjusting for other risk factors. 63 Hearing aid use was the largest factor protecting from decline (regression coefficient β for higher episodic memory 1·53; p<0·001) adjusting for protective and harmful factors. The long follow-up times in these prospective studies suggest hearing aid use is protective, rather than the possibility that those developing dementia are less likely to use hearing aids. Hearing loss might result in cognitive decline through reduced cognitive stimulation.
Traumatic brain injury (TBI)
The International Classification of Disease (ICD) defines mild TBI as concussion and severe TBI as skull fracture, oedema, brain injury or bleed. Single, severe TBI is associated in humans, and mouse models, with widespread hyperphosphorylated tau pathology, and mice with APOE ε4 compared to APOE ε3 allele have more hippocampal hyper-phosphorylated tau after TBI. 64 , 65 TBI is usually caused by car, motorcycle, and bicycle injuries; military exposures; boxing, horse riding, and other recreational sports; firearms; and falls. 66 A nationwide Danish cohort study of nearly 3 million people aged 50 years or older, followed for a mean of 10 years, found an increased dementia (HR 1·2, 95% CI 1·2–1·3) and Alzheimer's disease risk (1·2, 1·1–1·2). 67 Dementia risk was highest in the 6 months after TBI (4·1, 3·8–4·3) and increased with number of injuries in people with TBI (one TBI 1·2, 1·2–1·3; ≥5 TBIs 2·8, 2·1–3·8). Risk was higher for TBI than fractures in other body areas (1·3, 1·3–1·3) and remained elevated after excluding those who developed dementia within 2 years after TBI, to reduce reverse causation bias. 67
Similarly, a Swedish cohort of over 3 million people aged 50 years or older, found TBI increased 1-year dementia risk (OR 3·5, 95% CI 3·2–3·8); and risk remained elevated, albeit attenuated over 30 years (1·3, 1·1–1·4). 68 ICD defined single mild TBI increased the risk of dementia less than severe TBI and multiple TBIs increased the risk further (OR 1·6, 95% CI 1·6–1·7 for single TBI; 2·1, 2·0–2·2 for more severe TBI; and 2·8, 2·5–3·2 for multiple TBI). A nested case control study of early onset clinically diagnosed Alzheimer's disease within an established cohort also found TBI was a risk factor, increasing with number and severity. 69 A stronger risk of dementia was found nearer the time of the TBI, leading to some people with early-onset Alzheimer's disease.
Military veterans have a high risk of occupational TBI, and formal record keeping allows long-term follow-up. A study of 178 779 veterans with TBI with propensity-matched veterans without TBI found dementia risk was associated with TBI severity (HR 2·4, 95% CI 2·1–2·7 for mild TBI without loss of consciousness; 2·5, 2·3–2·8 for mild TBI with loss of consciousness; and 3·8, 3·6–3·9 for moderate to severe TBI). 70 Similarly women veterans with TBI had increased risk of dementia compared to those without TBI (1·5, 1·0–2·2). 71
A cohort study of 28 815 older adults with concussion, found the risk of dementia doubled, with 1 in 6 developing dementia over a mean follow-up of 3·9 years, although those taking statins had a 13% reduced risk of dementia compared to those who were statin-free. They suggest future RCTs as statins might mitigate injury-related brain oedema, oxidative stress, amyloid protein aggregation, and neuroinflammation. 72
The term chronic traumatic encephalopathy describes sports head injury, which is not yet fully characterised and covers a broad range of neuropathologies and outcomes, with current views largely conjecture. 73 The evidence has subsequently been strengthened by a study on Scottish former soccer players reporting that they are more likely than controls to have Alzheimer's disease specified on their death certificates (HR 5·1, 95% CI 2·9–8·8) and to have been prescribed any dementia-related medications (OR 4·9, 95% CI 3·8–6·3) but not on medical records. 74 The study controlled for socio-economic class based on residential address, which in footballers might be less linked to level of education.
Hypertension
Persistent midlife hypertension is associated with increased risk of a late life dementia. In the Framingham Offspring cohort comprising 1440 people, elevated systolic blood pressure (≥140 mm Hg in midlife; mean age 55 years) was associated with an increased risk of developing dementia (HR 1·6, 95% CI 1·1–2·4) over an 18 year follow-up period. 12 In this study risk increased further if hypertension persisted into later life (mean age 69 years; HR 2·0, 95% CI 1·3–3·1). In the same cohort, people in late midlife (mean age 62 years) with ideal cardiovascular parameters (current non-smoker, body mass index [BMI] 18·5–25 kg/m 2 , regular physical activity, healthy diet, optimum blood pressure <120/<80 mm Hg, cholesterol, and normal fasting blood glucose) were compared to people with at least one of these risks. 75 Those with ideal cardiovascular parameters had a lower 10-year risk of all-cause dementia (HR 0·8, 95% CI 0·1–1·0), vascular dementia (0·5, 0·3–0·8) and clinically diagnosed Alzheimer's disease (0·8, 0·6–1·0). In a UK cohort study of 8639 civil servants, a single measure of systolic blood pressure of 130 mm Hg or higher at age 50 years but not at age 60 or 70 years was associated with increased risk of dementia (1·4, 1·1–1·7). 13 In those with persistent systolic blood pressure of 130 mm Hg or higher, from mean age 45 to 61 years, dementia risk is increased even if free of cardiovascular disease relative to those without hypertension (1·3, 1·0–1·7).
A further cohort study has provided potential insights into mechanisms, reporting that midlife hypertension, defined as from age 40 years, was associated with reduced brain volumes and increased white matter hyperintensity volume but not amyloid deposition. 76 Of note, blood pressure declines in later life and this decline is associated with and, potentially caused by, dementia development (HR 2·4, 95% CI 1·4–4·2). 12 , 13 , 77
Antihypertensive drugs, aspirin, and statins
The US and Puerto Rico Systolic Blood Pressure Intervention Trial (SPRINT) in 9361 hypertensive adults aged 50 years and older, was stopped early because of significantly fewer cardiovascular events and deaths occurring in the intensive treatment arm (aiming for systolic <120 mm Hg, n=4678) in comparison with standard treatment (systolic <140 mm Hg, n=4683). 78 Cognitive assessment continued after stopping the trial intervention in SPRINT MIND. 79 In the intensive compared with the standard treatment group, there were 7·2 dementia cases as opposed to 8·6 cases/1000 person-years (HR 0·8; 95% CI 0·7–1·0) within on average 2 years from the end of the intervention period and 5 years after baseline. Pre-specified secondary outcomes were also reduced in the intensive arm for mild cognitive impairment (14·6 vs 18·3 cases/1000 person-years; HR 0·8, 95% CI 0·7–1·0), combined mild cognitive impairment or dementia (20·2 vs 24·1 cases/1000 person-years; HR 0·9, 95% CI 0·7–1·0) 79 making this the first trial to suggest reduction of risk for mild cognitive impairment. Those who were lost to follow-up were at greater risk of dementia than those who continued but follow-up rates did not differ according to intervention group. 80
Four meta-analyses of blood pressure medications to lower high blood pressure with six studies overlap have provided combined estimates of effects. All meta-analyses suggest reduced dementia in those in the interventions arms for outcomes of any dementia as well as clinically diagnosed Alzheimer's disease. The first included randomised controlled trials (RCTs) of any drug to lower blood pressure and reported a reduction in risk of around 10% at marginal significance (RR 0·9, 95% CI 0·9–1·0). 81 Meta-regression showed risk lowered more if the achieved systolic pressure differential was larger between the intervention and control group. The second included 15 trials and observational studies of diuretics involving 52 599 people (median age 76 years) with 6·1 years median follow-up (dementia HR 0·8, 95% CI 0·8–0·9 and Alzheimer's disease 0·8, 0·7–0·9). 82 The third included used individual participant data from six observational studies; (dementia 0·9, 0·8–1·0 and Alzheimer's disease 0·8, 0·7–1·0; figure 4 ). 83 The fourth focused on people prescribed calcium channel blocker only, included 10 RCTs and observational studies comprising 75 239 hypertensive older adults (median age 72 years, median follow-up 8·2 years) found lowered dementia risk (RR 0·7, 95% CI 0·6–0·9). 84 A 2019 meta-analysis addressing which class of anti-hypertensive drug to use to lower risk of either incident dementia or cognitive decline, found over 50 000 participants in 27 studies and reported no consistent difference in effect according to which class of drug was used. 85
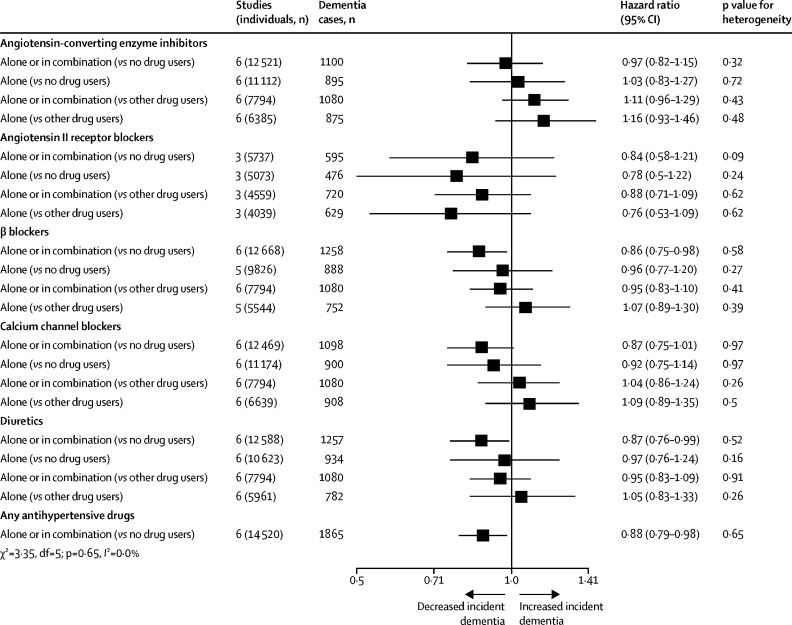
Associations of antihypertensive medication use with incident dementia in those with high blood pressure
Adapted from Ding et al, 83 by permission of Elsevier.
A Cochrane review reported good evidence that statins given to older people at risk of vascular disease do not prevent cognitive decline or dementia. 86 One RCT found 100 mg aspirin versus placebo in 19 114 healthy adults older than 65 years did not reduce dementia (HR 1·0, 95% CI 0·8–1·2), death, physical disability, or cardiovascular disease over a period of 4·7 years. 87
Physical inactivity, exercise, and fitness
Studies of physical activity are complex. Patterns of physical activity change with age, generation, and morbidity and are different across sex, social class, and cultures. The studies suggest a complicated relationship with the potential for both risk reduction and reverse causation.
Meta-analyses of longitudinal observational studies of 1–21 years duration showed exercise to be associated with reduced risk of dementia. 2 A further overview of systematic reviews concluded that there is convincing evidence for physical activity protecting against clinically diagnosed Alzheimer's disease. 88
Since the 2017 Commission, the HUNT study of 28 916 participants aged 30–60 years has been published, reinforcing the previous literature in this area. At least weekly midlife moderate-to-vigorous physical activity (breaking into a sweat) was associated with reduced dementia risk over a 25-year period of follow-up (HR 0·8, 95% CI 0·6–1·1) but the confidence intervals were wide. 89 In contrast the Whitehall Study reporting on the 28-year follow-up of 10 308 people, found that more than 2·5 hours of self-reported moderate-to-vigorous physical activity per week, lowered dementia risk over 10, but not 28 years. 33 Very long-term studies are unusual; however, one 44-year study recruited 191 women (mean age 50) purposively to be representative of the Swedish population and reported that 32% of the participants with low baseline peak fitness, 25% with medium, and 5% with high fitness developed dementia (high vs medium HR 0·1, 95% CI 0·03–0·5, low vs medium 1·4, 0·7–2·8). 90
An individual-level meta-analysis of 19 observational studies of relatively younger adults included 404 840 participants' data (mean baseline age 45·5 years; mean follow-up duration 14·9 years), reporting an increased incidence of all-cause dementia (HR 1·4, 95% CI 1·2–1·7) and clinically diagnosed Alzheimer's disease (1·4, 1·1–1·7) in those who were physically inactive in the 10-year period before diagnosis. 91 Notably, however, no difference in dementia risk measured 10–15 years before time of dementia incidence was found except in those with comorbid cardio-metabolic disease (RR 1·3, 95% CI 0·8–2·1).
People might stop exercising due to prodromal dementia so inactivity might be either a consequence or a cause or both in dementia and might be more of a risk in those with cardiovascular morbidity. As with other outcomes, exercise might be required to be sustained and continue nearer the time of risk. 92
Trials of exercise
Since the 2017 Commission several meta-analyses and systematic reviews have been published with three high quality meta-analyses which we include. The first included 39 RCTs with an unclear total number of participants examining moderate or vigorous exercise of any frequency lasting 45–60 min per session in cognitively normal adults aged older than 50 years. This analysis reported global cognitive improvements (standard mean difference [SMD]=0·3, 95% CI 0·2–0·4) for moderate or vigorous resistance (13 studies) or aerobic exercise (18 studies) lasting 45–60 min per session with no difference between them but no effect found for yoga. 93 A second meta-analysis of RCTs in people with mild cognitive impairment found global cognition improved in the intervention group (0·3, 0·1–0·5) with aerobic exercise having a higher effect (0·6, 0·5–0·6). 94 This study did not have dementia as an outcome measure. A third meta-analysis of RCTs of longer term exercise found five studies (four lasting 12 months and one 24 months) with 2878 participants with normal baseline cognition. 95 The incidence of dementia was 3·7% (n=949) for exercisers and 6·1% (n=1017) for controls (random effect RR 0·6, 95% CI 0·3–1·1; fixed effect as no evidence of heterogeneity 0·7, 0·4–1·0). The authors concluded that the study showed no significant effect of exercise for reducing dementia, mild cognitive impairment, or clinically significant cognitive decline but was underpowered. WHO guidelines have been published since the 2017 Commission, suggesting specific activity levels drawing on these, and one further systematic review which considered sex differences on the effect of exercise. 96 , 97 It concluded the evidence points towards physical activity having a small, beneficial effect on normal cognition, with a possible effect in mild cognitive impairment, mostly due to aerobic exercise. 97 Evidence about the effect of specific types of exercise, such as progressive muscle resistance training, on dementia risk is scarce.
In the 2017 Commission we reported on diabetes as a risk factor for dementia. Distinguishing between treated and untreated diabetes as a risk factor for dementia is challenging in observational studies. In a pooled meta-analysis from over 2·3 million individuals with type 2 diabetes across 14 cohort studies, including 102 174 with dementia, diabetes was associated with an increased risk of any dementia (RR 1·6, 95% CI 1·5–1·8 for women and 1·6, 1·4–1·8 for men). 98 The risk of dementia increased with the duration and severity of diabetes. The effect of different diabetic medications on cognition or dementia outcomes remains unclear as few studies have investigated this area. 99 However, one meta-analysis of cohort studies of diabetes reported that, cross sectionally, people with diabetes taking metformin had lower prevalence of cognitive impairment (three studies OR 0·6, 95% CI 0·4–0·8) and, longitudinally, reduced dementia incidence (six studies HR 0·8, 95% CI 0·4–0·9) compared with those taking other medications or no medication. 100 However another analysis did not find a protective effect of metformin for incident dementia (three studies, RR 1·1, 95% CI 0·5–2·4) with possible harm with insulin therapy (1·2, 1·1–1·4); but this did not account for severity of diabetes of those with type 2 diabetes on insulin. 99 A Cochrane review reported intensive compared to standard diabetes control trials with 5 year follow up (n=11 140), showing no impact on cognitive decline (1·0, 95% CI 0·9–1·1) or dementia (1·3, 0·9–1·9). 101
Overall type 2 diabetes is a clear risk factor for development of future dementia; however, whether any particular medication ameliorates this risk is unclear. Intensive diabetic control does not decrease the risk of dementia.
Combined cardiovascular risk factors
Studies of individual cardiovascular risk factors usually control for other cardiovascular risks, which cluster in individual people. This does not take into account the combinations and contexts in which risk occurs. A UK study of 7899 people aged 50 years followed up for 25 years, calculated a cardiovascular health score based on four behaviour-related (smoking, diet, physical activity, BMI) and three biological (fasting glucose, blood cholesterol, blood pressure) metrics each coded on a three-point scale (0, 1, 2). 100 A better score was associated with a lower risk of dementia (HR 0·9, 95% CI 0·9–1·0 per 1 point scale increment), for both behaviour-related (HR/1 point increment in subscales 0·9, 95% CI 0·8–0·9) and biological subscales (0·9, 0·8–1·0), maintained in people free of cardiovascular disease over the follow-up (0·9, 95% CI 0·8–1·0). These authors also reported an association of the score on the scale with hippocampal atrophy and total brain volume but not white matter hyperintensities. This finding underlines the importance of clustering of cardiovascular risk factors in midlife, as studies of individual risk factors in this sample had not shown a significant association, when controlling for other individual risks. 33
Excessive alcohol consumption
Heavy drinking is associated with brain changes, cognitive impairment, and dementia, a risk known for centuries. 102 An increasing body of evidence is emerging on alcohol's complex relationship with cognition and dementia outcomes from a variety of sources including detailed cohorts and large-scale record based studies. Alcohol is strongly associated with cultural patterns and other sociocultural and health-related factors, making it particularly challenging to understand the evidence base.
A French 5-year longitudinal study of over 31 million people admitted to hospital, found alcohol use disorders (harmful use or dependence as defined in ICD) were associated with increased dementia risk, calculated separately for men and women (women HR 3·3, 95% CI 3·3–3·4, men 3·4, 3·3–3·4). 103 The relationship of dementia with alcohol use disorders was particularly clear in the earlier onset dementias (age less than 65 years) in which 56·6% had an alcohol use disorder noted in their records (n=57 353; 5·2% all dementias).
A systematic review incorporating 45 studies of light to moderate drinking using a variety of definitions reported a reduced risk of dementia compared with not drinking (RR 0·7; 95% CI 0·6–0·91). 104 Risk was not reported separately for men and women. Drinking less than 21 units of alcohol per week (1 unit of alcohol=10 mL or 8 g pure alcohol) might be associated with a lower risk of dementia. 105 , 106 A 5-year follow-up study of 13 342 men and women volunteers from UK biobank aged 40–73 years who drank, included few heavy drinkers and did not analyse abstainers. 106 The study reported that those who drank more than 12 units per week declined slightly more in reaction time in a perceptual matching task than those who drank less (β2=−0·07, 95% CI −0·09 to −0·04). 106 The UK Whitehall study with 23 years follow-up, included 9087 participants aged 35–55 years at baseline. 107 Drinking more than 21 units per week and long-term abstinence were both associated with a 17% (95% CI 4–32 and 13–23 respectively) increase in dementia compared to drinking less than 14 units. Drinking more than 14 units was also associated with right sided hippocampal atrophy on MRI. 108
Weight control and obesity
Overweight is an emerging concern, given the changing BMI across the world's ageing population. New evidence supports the relationship between increased BMI and dementia from a review of 19 longitudinal studies including 589 649 people aged 35 to 65 years, followed up for up to 42 years. It reported obesity (BMI ≥30; RR 1·3, 95% CI 1·1–1·6) but not being overweight (BMI 25–30; 1·1, 1·0–1·2) was associated with late-life dementia. 109 In a further meta-analysis of individual level data from 1·3 million adults (aged ≥18 years), which included two studies from the meta-analysis cited above, 109 higher body mass measured before probable preclinical and prodromal dementia was associated with increased dementia risk (RR 1·3, 1·1–1·7/5-unit increase in BMI). 11
Weight loss in midlife and dementia risk
A meta-analysis of seven RCTs (468 participants) and 13 longitudinal studies (551 participants) of overweight and obese adults without dementia, mean age 50 years, found weight loss of 2 kg or more in people with BMI greater than 25 was associated with a significant improvement in attention and memory. All but one of the studies included participants aged younger than 65 years. The RCTs reported memory improvement over 8–48 weeks (SMD=0·4, 95% CI 0·2–0·6) and short-term longitudinal studies found improvement over a median of 24 weeks (SMD=0·7, 95% CI 0·5–0·8); however, data about the long-term effects or the effect of weight loss in preventing dementia are absent. 110
Smokers are at higher risk of dementia than non-smokers, 2 and at a higher risk of premature death before the age at which they might have developed dementia, introducing some bias and uncertainty in the association between smoking and risk of dementia. 111 , 112 Stopping smoking, even when older, reduces this risk. Among 50 000 men aged older than 60 years, stopping smoking for more than 4 years, compared to continuing, substantially reduced dementia risk over the subsequent 8 years (HR 0·9; 95% CI 0·7–1·0). 113 Worldwide, 35% of non-smoking adults and 40% of children are estimated to be exposed to second-hand smoke; 114 although literature on the impact of this exposure and dementia risk is scarce. One study indicated that in women aged 55–64 years, second-hand smoke exposure was associated with more memory deterioration and the risk increased with exposure duration even after controlling for other confounding factors. 115
Depression is associated with dementia incidence, with a variety of possible psychological or physiological mechanisms. It is also part of the prodrome and early stages of dementia. Reverse causation is possible whereby depressive symptoms result from dementia neuropathology that occurs years before clinical dementia onset. These explanations are not mutually exclusive. As in diabetes, few studies considering depression as a risk factor for dementia have distinguished between treated and untreated depression. In a meta-analysis of 32 studies, with 62 598 participants, with follow-up from 2 to 17 years, a depressive episode was a risk factor for dementia (pooled effect size 2·0, 95% CI 1·7–2·3). 116 Meta-regression analysis revealed a non-significant trend for the association between depression and incident dementia to be weaker when the length of follow-up was longer. The Norwegian HUNT study, suggested that symptoms of psychological distress predicted dementia 25 years later however with wide bounds of uncertainty (HR 1·3, 95% CI 1·0–1·7). 89 Two further studies differentiate between late-life and earlier life depressive symptoms. The UK Whitehall study, in a follow-up of 10 189 people, reports that in late life these symptoms increase dementia risk but not at younger ages (follow-up 11 years HR 1·7; 95% CI 1·2–2·4; follow-up 22 years 1·0, 0·7–1·4). 34 , 117 A 14-year longitudinal study of 4922 initially cognitively healthy men, aged 71–89 years, found depression was associated with 1·5 (95% CI 1·2- 2·0) times the incidence of dementia but this association was accounted for by people developing dementia within 5 years of depression. 118 The use of antidepressants did not decrease this risk.
A study of 755 people with mild cognitive impairment and with a history of depression from the Australian longitudinal Alzheimer's Disease Neuroimaging Initiative, considered the effect of selective serotonin-reuptake inhibitor (SSRI) treatment, such as citalopram, known to reduce amyloid plaque generation and plaque formation in animal models. 119 The study found that more than 4 years of such treatment was associated with delayed progression to clinically diagnosed Alzheimer's disease. People treated with antidepressants seem likely to differ from those who are not treated. Thus, the question of whether antidepressant treatment mitigates dementia risk remains open.
Social contact
Social contact, now an accepted protective factor, enhances cognitive reserve or encourages beneficial behaviours, although isolation might also occur as part of the dementia prodrome. Several studies suggest that less social contact increases the risk of dementia. Although most people in mid and later life are married, by the time they reach older age, disproportionate numbers of women are widowed as they outlive their husbands, thus reducing their social contact. In these generations, marital status is therefore an important contributor to social engagement. Additionally, most marriages are in the relatively young, and married people usually have more interpersonal contact than do single people—this gives a long-term estimate of the effect of social contact. A systematic review and meta-analysis including 812 047 people worldwide found dementia risk to be elevated in lifelong single (RR 1·4, 95% CI 1·1–1·9) and widowed people (1·2, 1·0–1·4), compared with married people and the association was consistent in different sociocultural settings. 120 Studies adjusted for sex and we do not know if a differential risk between men and women exists. Differences persisted in studies that adjusted for education and physical health so might be attributable to married people having more social contact, rather than solely because they tend to have better physical health and more education, although residual confounding is possible. A systematic review and meta-analysis of 51 longitudinal cohort studies of social isolation and cognition included 102 035 participants aged 50 or more years at baseline, with follow-up of 2–21 years. 121 High social contact (measured through either or both of social activity and social network) was associated with better late-life cognitive function (r=0·05, 95% CI: 0·04–0·065) and no differences according to sex or length of time followed up.
A new meta-analysis found that in long-term studies (≥10 years), good social engagement was modestly protective (n=8876, RR=0·9, 95% CI 0·8–1·0); but loneliness was not associated with dementia risk. 122 No long term (>10 years) studies of loneliness and dementia outcomes have been done.
A UK 28-year follow-up study of 10 308 people found that more frequent social contact at age 60 years was associated with lower dementia risk over 15 years of follow-up (HR for one standard deviation social contact frequency 0·9, 95% CI 0·8–1·0). This finding suggests more frequent social contact during late middle age is associated with a modest reduction in dementia risk, independent of socio-economic and other lifestyle factors. 123 A Japanese longitudinal cohort study of 13 984 adults aged older than 65 years with a mean of 10 years follow-up calculated a five-point social contact scale based on: marital status; exchanging support with family members; having contact with friends; participating in community groups; and engaging in paid work. It found the score to be linearly associated with reduced dementia risk; those who scored highest on the five-point scale were 46% less likely to develop incident dementia compared with those in the lowest category. 124
Despite clear cultural variation in the meaning and perception of social isolation, findings of protective effect of more social contact are largely consistent in different settings and for either sex across the studies and meta-analyses. 118 , 120 , 121
Social interventions
Little evidence of the effects of social interventions on dementia exists but a systematic review of low quality RCTs of 576 adults aged 60 or more years with normal cognition found facilitated meeting and discussion groups were associated with improved global cognition and increased brain volume at follow-up. 118
Air pollutants
Air pollution and particulate pollutants are associated with poor health outcomes, including those related to non-communicable diseases. Attention has turned to their potential effect on the brain. Animal models suggest airborne particulate pollutants accelerate neurodegenerative processes through cerebrovascular and cardiovascular disease, Aβ deposition, and amyloid precursor protein processing. 125 , 126 Although the higher levels of dementia from air pollutants are still subject to the potential for residual confounding, the effects on animal models are evidence of physiological effects over and above those driven by life-course deprivation.
High nitrogen dioxide (NO 2 ) concentration (>41·5 μg/m 3 ; adjusted HR 1·2, 95% CI 1·0–1·3), fine ambient particulate matter (PM) 2·5 from traffic exhaust (1·1, 1·0–1·2) 127 , 128 , 129 and PM 2·5 from residential wood burning (HR=1·6, 95% CI 1·0–2·4 for a 1 μg/m 3 increase) are associated with increased dementia incidence. Traffic often produces NO 2 and PM 2·5 and it is hard to separate their effects, although evidence for additive effects of different pollutants exists. 127 , 128 , 129 A systematic review of studies until 2018 including 13 longitudinal studies with 1–15 years follow-up of air pollutants exposure and incident dementia, found exposure to PM 2·5, NO 2 , and carbon monoxide were all associated with increased dementia risk. 24 The attributable burden of dementia and excess death from PM 2·5 in one large 10-year US study was particularly high in Black or African American individuals and socio-economically disadvantaged communities and related to particulate PM 2·5 concentrations above the US guidelines. 130
Mechanisms by which sleep might affect dementia remain unclear, but sleep disturbance has been linked with β-amyloid (Aβ) deposition, 131 , 132 reduced glymphatic clearance pathways activation, 133 low grade inflammation, increased Tau, hypoxia 132 , 134 and cardiovascular disease. 135 Sleep disturbance is hypothesised to increase inflammation which raises Aβ burden, leading to Alzheimer's disease and further sleep disturbance. 136
Two meta-analyses showed similar findings. The first was a synthesis of longitudinal studies with an average of 9·5 years follow-up and the second reported cross-sectional and prospective cohort studies of mixed quality with different methods of measuring sleep. Sleep disturbances were defined broadly, often self-reported and including short and long sleep duration, poor sleep quality, circadian rhythm abnormality, insomnia, and obstructive sleep apnoea. All these disturbances were associated with a higher risk of all-cause dementia (RR 1·2; 95% CI 1·1–1·3) 137 and clinically diagnosed Alzheimer's disease (1·6, 1·3–1·9) compared with no sleep disturbance, although not all cohort studies excluded those with cognitive impairment or dementia at baseline from their analyses. 138 A U-shaped association has been reported between sleep duration and risk of mild cognitive impairment or dementia with higher risks of dementia with less than 5 hours (HR=2·6; 95% CI 1·4–5·1) compared with more than 5 and less than 7 and more than 10 hours sleep (2·2, 1·4–3·5) and risks for all-cause dementia and clinically diagnosed Alzheimer's disease being similar. 135 , 139 , 140 , 141
The postulated mechanisms of reduced sleep leading to accumulation of Alzheimer's type pathology is inconsistent with the evidence that both more sleep and less sleep are associated with increased risk of dementia. New onset late-life sleep disturbance, a few years before clinical dementia, might be part of the natural history of the dementia syndrome, appearing to be a risk factor, or reflect other disorders, for example, mood disturbances or cardiovascular disease. 135 , 142 Hypnotic use might increase risks although this is unclear and a 2018 study 139 suggests that findings of a connection were related to reverse causality and confounders. 143 When benzodiazepine use was considered, in one study, sleep length was no longer significant 139 but not in all studies. 135 Those taking hypnotics were at greater risk of dementia than those who did not regardless of sleep duration. 139 Medication for sleep disturbance might be harmful and benzodiazepines are associated with falls, hospital admissions, and possibly dementia. 139 , 144
Nutrition and dietary components are challenging to research with controversies still raging around the role of many micronutrients and health outcomes in dementia. Observational studies have focused on individual components ranging from folate and B vitamins, Vitamin C, D, E, and selenium amongst others as potential protective factors. 88 There has been a move towards considering the evidence base for whole diets in the last 5 years, particularly high plant intake such as in the Mediterranean diet (high intake of vegetables, legumes, fruits, nuts, cereals, and olive oil; low intake of saturated lipids and meat) or the similar Nordic diet, rather than individual nutrients, which might reduce cognitive decline and dementia. 145 One example is a longitudinal cohort study of 960 participants, ages 58–99 years, in which those reporting the highest intake of green leafy vegetables, equivalent to 1·3 servings per day, had less cognitive decline over 4·7 years than those reporting the lowest intake (β=0·05 standardised units 95% CI 0·02–0·07). 146 The authors report this difference as being equivalent to being 11 years younger. A further prospective cohort study with three midlife dietary assessments in 8255 people, followed up for a mean of nearly 25 years, found neither healthy dietary pattern nor Mediterranean diet protected from dementia, except in those with cardiovascular disease, suggesting that diet might influence dementia risk by protecting from the excess risk of cardiovascular risk factors. 147
Dietary interventions
As well as whole diets, there has been some interest in multi-nutrient interventions. A systematic review and a Cochrane review including RCTs of supplements (A, B, C, D, and E; calcium, zinc, copper, and multivitamins trials, n-3 fatty acids, antioxidant vitamins, and herbs) found a lack of evidence for supplement use to preserve cognitive function or prevent dementia in middle-aged (45–64 years) or older people (aged 65 years and older). 148 , 149 Cochrane reviews found no evidence for beneficial effects on cognition of those with mild cognitive impairment of supplementation with B vitamins for 6 to 24 months 150 or with vitamin E in preventing progression from mild cognitive impairment to dementia. 151 A 24-month RCT of 311 people of a multi-nutrient drink containing docosahexaenoic acid, vitamins B12, B6, folic acid, and other nutrients; found no significant effect on preventing cognitive deterioration in prodromal Alzheimer's disease. 152 The authors comment that the control group's cognitive decline was much lower than expected, leading to an inadequately powered trial.
Meta-analysis of two RCTs with 471 participants with normal cognition found the Mediterranean diet improved global cognition compared to controls (SMD 0·2, 95% CI 0·0–0·4). 153 A further meta-analysis identified five RCTs (n=1888) with a weak effect on global cognition (SMD 0·2, 95% Cl 0·0–0·5) 154 but no benefit of Mediterranean diet for incident cognitive impairment or dementia.
The WHO guidelines recommend a Mediterranean diet to reduce the risk of cognitive decline or dementia, as it might help and does not harm, but conclude Vitamins B and E, polyunsaturated fatty acid, and multicomplex supplementation should not be recommended. 97
Trials of combination strategies to prevent dementia
The FINGER RCT was a 2-year multidomain intervention to prevent cognitive decline and dementia in 1260 people with cardiovascular risk factors aged 60–77 years, recruited from a Finnish national survey. Similar multidomain studies were discussed in the 2017 Commission. 2 FINGER found a small group reduction in cognitive decline in the intervention group compared with control (comprehensive neuropsychological test battery Z score 0·02, 95% Cl 0·00–0·04) regardless of baseline sociodemographic, socio-economic, cognitive, or cardiovascular status. 155 However, in a subgroup analysis, greater beneficial effects were observed on processing speed in individuals with higher baseline cortical thickness in Alzheimer's disease areas. 156
The Healthy Ageing Through Internet Counselling in the Elderly (HATICE) study recruited 2724 older people (≥65 years) in the Netherlands, Finland, and France with two or more cardiovascular risk factors. 157 , 158 It compared an interactive internet platform plus remote support by a coach, aiming to improve self-management of vascular risk factors, with a non-interactive control platform with basic health information. A small improvement in the cardiovascular risk composite primary outcome was observed in the intervention group compared with the control group at 18 months, mainly through weight loss, and the dementia risk score was slightly lower in those who received the intervention (mean difference −0·15, 95% CI −0·3 to −0·0). A larger effect was observed in the younger age group (65–70 years) and those with the lowest level of education, who had a higher baseline risk, suggesting that targeting high-risk populations might be more effective. Several multidomain preventive trials are ongoing—for example, World Wide FINGERS .
Total PAF calculation
We incorporated excessive alcohol consumption, TBI, and air pollution into our life-course model of dementia, as well as the original nine risk factors, because of the updated evidence. To calculate new RRs for excessive alcohol consumption, TBI and air pollution, we systematically reviewed the literature and did new meta-analyses for excessive alcohol consumption and TBI. For the other nine factors, we used values for RR and risk factors prevalence from our previous analysis and calculated communality using the same method as in the 2017 Commission. 2
PAF calculation
We used a representative sample of over 10 000 UK community-dwelling adults, to calculate communality (clustering of risk factors) of 11 risk factors for which data existed, 159 to allow calculation of each factor's unique risk. As we could find no datasets measuring TBI, with the other 11 risk factors of interest, we could not calculate its communality. We therefore used the mean of the other 11 communalities to calculate a weighted PAF, so we could include TBI. We used cohabitation as a proxy measure for social contact, and urbanicity for air pollution exposure. Our analysis found four principal components, explaining 55% of the total variance between the eleven risk factors, suggesting substantial overlap. The appendix (p 2) shows the PAF formula and the steps in calculating communality and we detail our new meta-analyses next, which we used to update the figure and perform our new calculations.
Incorporation of the new chosen risks in new systematic reviews
We searched, from inception to Oct 29, 2019, Embase, Allied, and Complementary Medicine, MEDLINE, and PsycINFO terms “dementia” OR “dement*” OR “AD” OR “VaD”, “Alzheimer*” AND “alcohol” OR “ethanol” OR “alcohol*” OR “drink*” OR “drunk*” to update an earlier review. 160 We used inclusion criteria: original population-based cohort studies measuring drinking during midlife, as alcohol intake tends to fall with age; 161 alcohol consumption quantified at baseline by units or number of drinks (one drink, 1·5 units) per week; and all-cause dementia ascertained at follow-up using validated clinical measures. We contacted authors for additional data. 162 Three studies met our inclusion criteria. 107 , 162 , 163 We converted HRs to RRs 164 and used raw data 162 to calculate RR, 165 for our random effects meta-analysis using Generic Inverse Variance Methods. The RR associated with drinking—more than 21 units (168 g) of alcohol weekly—compared with lighter drinking was 1·18 (95% Cl 1·06–1·31; figure 5 ). We used Health Survey England figures for heavier drinking prevalence to calculate PAF as we could not find a worldwide estimate. The weighted PAF was 0·8.
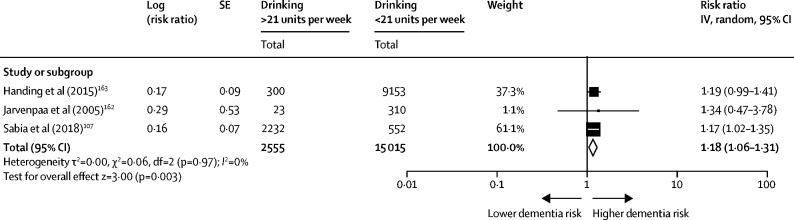
Meta-analysis of relative risk of dementia associated with drinking more than 21 units of alcohol per week in midlife compared to lighter consumption of alcohol
To estimate the RR of TBI of all severities for all cause dementia, we searched Embase, Medline, and PsycINFO from Jan 1, 2016, to Oct 21, 2019, updating an earlier search, 166 using terms (“traumatic brain injury” or “head injury” or “brain injury” or TBI) AND (neurodegeneration or “cognitive dysfunction” or dementia or “Alzheimer's disease” or “Parkinson's disease” or “frontotemporal dementia”). We converted HR figures to RR. 164 , 167 We used inclusion criteria: original population-based cohort studies, baseline TBI of all severities reported, and all-cause dementia ascertained at follow-up using validated clinical measures. We combined four new studies meeting inclusion criteria 67 , 68 , 71 , 168 with the four studies meeting criteria from the original review in a random effects meta-analysis. 166 The pooled RR was 1·84 (95% CI 1·54–2·20) for all cause dementia from all severities of TBI ( figure 6 ) although there was heterogeneity in study-specific estimates, possibly because of different populations. We used the TBI adult population prevalence of 12·1% from a meta-analysis to calculate PAF. 173 The weighted PAF was 3·4.
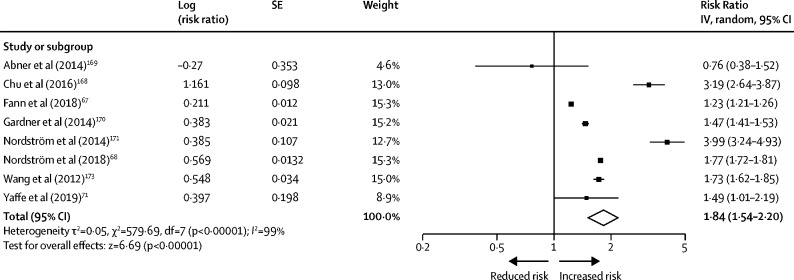
Meta-analysis of relative risk of all-cause dementia associated with all severity midlife traumatic brain injury
A 2019 systematic review synthesised observational studies, finding consistently increased risk of dementia from air pollution, but heterogeneous comparator groups precluded meta-analysis. 24 We updated the search, using the same search terms and searching MEDLINE, Embase, and PsycINFO from Sept 20, 2018, (the end date of the last search) to Oct 22, 2019. We included longitudinal studies with assessment of all cause air pollution exposure; use of formal assessment of cognitive function at baseline; report of incident all-cause dementia, data from adults (age ≥18 years); and a minimum follow-up of 6 months. As meta-analysis was not possible, we used data from the only study of all-cause air pollution with the outcome of all-cause dementia, with low-moderate risk of bias. This population-based, observational cohort was from Canada, where pollutant concentrations are among the lowest in the world and examined 2 066 639 people, with a mean baseline age of 67 years. 174 We calculated the RR of dementia for those in the three highest quartiles compared to the lowest was 1·09 (1·07–1·11). The attributable fraction for exposure to the highest three quartiles versus the lowest quartile of PM 2·5 and NO 2 was 6·1% (4·8–7·5). The weighted PAF was 2·3.
Table 1 displays the prevalence, communality, relative risk, unweighted and weighted PAFs adjusted for communality. Figure 7 shows the updated life-course model of potentially modifiable risk factors for dementia, including the three new risk factors.
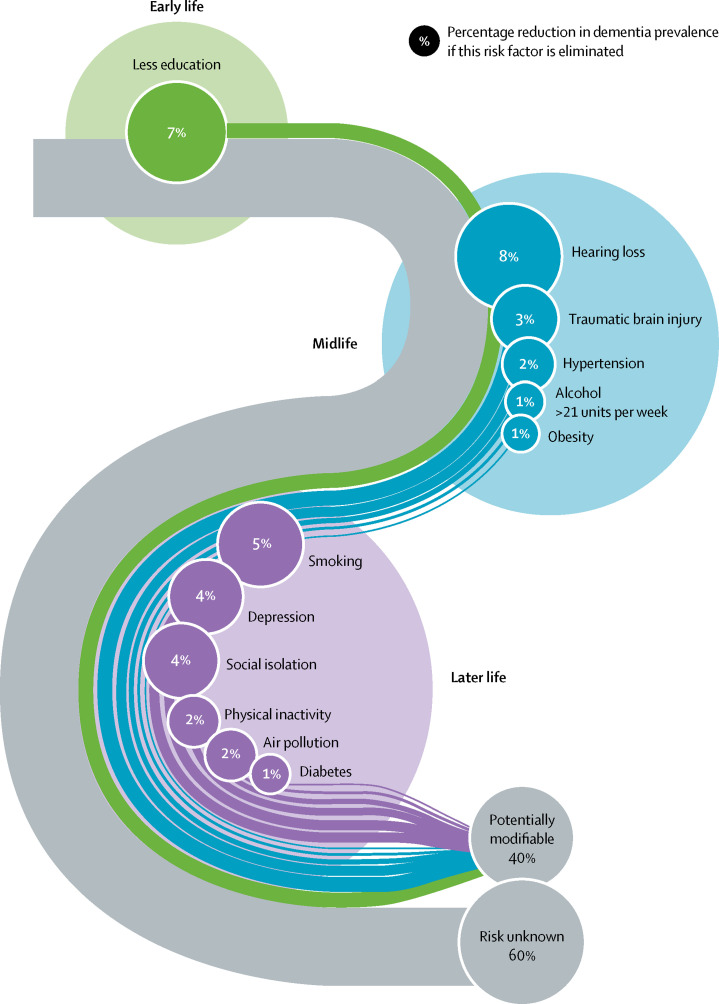
Population attributable fraction of potentially modifiable risk factors for dementia
Strengths and limitations
This Commission is the most comprehensive analysis to date and updates the 2017 Commission with emerging risk factor evidence convincing enough to calculate PAF for potentially reversible risk factors. We reviewed the literature systematically for the chosen risk factors and provided illustrative new literature to update our synthesis and identify data to calculate communality. We find a hopeful picture with an estimate of around 40% of all cases of dementia being associated with 12 potentially modifiable risk factors.
We have made assumptions to calculate this new model. We used global figures for dementia risk although we know the risk factors prevalence varies between countries and most global research is from HIC, so LMIC are under-represented because of lack of data. We have assumed a causal relationship between risk factors and dementia, although we have been cautious and not included risk factors with less good evidence. No single database exists with all 12 risk factors together, but we found 11 of the factors in a UK database and used the mean figure for communality calculations for TBI. We calculated communality for the other 11. We do not know how far findings of communality in other geographical populations might differ, or in those with a differing distribution of age groups or sex. We found that social isolation was not explicitly measured and had to use proxies, such as cohabitation when considering prevalence, which are approximate.
Specifically, evidence for the association of alcohol misuse with dementia comes from HIC and future studies from LMIC are needed to complete the picture. Exposure to air pollution changes over a lifetime and is inextricably linked to poverty and deprivation. However, the effects on animal models suggests specific physiological effects over and above those driven by life-course deprivation. We also considered the overlap with education for this and other risk factors and the correction for education, strongly inversely linked to deprivation, will address at least some of the confounding. However, the results in one study which reported the effect of air pollution on incident dementia showed very little difference in estimates before and after adjustment for education and other risk factors, suggesting little residual confounding exists. 174 We were also unable to meta-analyse data on pollution and thus unlike the other relative risks, the figure comes from only one study, from an area of low pollution so is likely to be an underestimate.
The longitudinal evidence linking potentially modifiable risk factors to dementia generally fulfils causality criteria in observational data (strength, consistency, biological plausibility, temporality, dose–response, coherence, and quasi-experimental studies, for example, more education or using hearing aids). When measuring a risk nearer to the age of dementia onset, then it is more likely that prodromal change affects, or even causes it. Alternatively, a risk factor might act on preclinical pathology or even cause dementia near the time of exposure. Thus, excessive alcohol, and TBI are particularly important in young-onset dementia, although many early onset dementias relate to genetic risks. Risk factors might also matter more at a time of higher biological vulnerability, which the studies we have drawn on cannot establish. The length of exposure required for risk or protection effect, and their inter-relationships as they change across life is unclear—it seems probable that longer or more intense exposure has stronger effects. Additionally, as our communality figures show, risk factors overlap. We cannot establish from these data if having multiple risk factors has an additive or synergistic effect. Association does not prove causation, however, as already noted, the reductions in prevalence and incidence in several HIC suggests that at least some of the risk factors estimated here do have a causal relationship with the clinical expression of dementia.
Key points and recommendations
We judge that sufficient new evidence supports adding three additional modifiable risk factors for dementia to our 2017 Commission model (excessive alcohol, traumatic brain injury, and air pollution). We have been able to add updated evidence on the nine risk factors implicated in the 2017 Commission (education, hypertension, hearing impairment, smoking, obesity, depression, inactivity, diabetes, and social contact). Reduction of these risk factors might be protective for people with or without a genetic risk, although study findings have not been entirely consistent. 175 , 176 , 177 , 178 As we noted in the 2017 Commission, others have previously calculated an estimate of the risk associated with APOε4 at 7% taking into account some other risk factors and this estimate highlights how relatively important potentially modifiable risk factors are in dementia. 2 , 179
For some risk factors, the pattern of risk and the individual's other health, both physical and mental, might be especially important. Currently, the evidence suggests a Mediterranean or Scandinavian diet might have value in preventing cognitive decline in people with intact cognition, particularly as one component of a healthy lifestyle, although how long the exposure has to be or during which ages is unclear. We do not recommend taking additional vitamins, oils, or mixed dietary supplements as a means of preventing dementia as extensive testing in trials has not led to signals of beneficial effects.
Data from RCTs on interventions to prevent cognitive decline, all-cause dementia, or Alzheimer's disease are few. For some key life influences, only observational data, particularly related to natural experiments such as changing the statutory education age, are possible. These influences should be investigated systematically wherever possible. Others can theoretically be investigated but the long follow-up required for midlife risk and protective factors and non-random attrition in longer studies are challenging. Using intermediate endpoints, such as cognition, and dementia onset in research remains uncertain because no intermediate markers with such a close relationship to dementia outcomes exist that it would be possible to predict with certainty for any given individual, age, and sex. Overall, the evidence for treating hypertension is strongest and high blood pressure throughout midlife increases the risk of dementia even without stroke.
Although a need for more evidence is apparent, recommendations should not wait, as clear indications of ways to reduce the chances of developing dementia without causing harm will also lead to other health and wellbeing benefits.
Our recommended strategies for dementia risk reduction include both population-wide and targeted interventions ( panel ). It is important to remember that more socially disadvantaged groups, including Black, Asian, and minority ethnic groups, are particularly at risk.
Panel. Recommended strategies for dementia risk reduction.
Risks are particularly high in more socially disadvantaged populations including in Black, Asian, and minority ethnic groups.
Population-wide
Prioritise childhood education for all, worldwide
Implement social public health policies that reduce hypertension risk in the entire population
Develop policies that encourage social, cognitive, and physical activity across the life course for all (with no evidence for any specific activities being more protective)
Scrutinise the risks for hearing loss throughout the life course, to reduce the risk of exposure to this risk factor
Reduce the risk of serious brain trauma in relevant settings, including occupational and transport
National and international policies to reduce population exposure to air pollution
Continue to strengthen national and international efforts to reduce exposure to smoking, both for children and adults, and to reduce uptake and encourage cessation
Targeted on individuals
Treat hypertension and aim for SBP <130 mm Hg in midlife
Use hearing aids for hearing loss; we need to help people wear hearing aids as many find them unacceptable, too difficult to use, or ineffective
Avoid or discourage drinking 21 or more units of alcohol per week
Prevent head trauma where an individual is at high risk
Stopping smoking is beneficial regardless of age
Reduce obesity and the linked condition of diabetes by healthy food availability and an environment to increase movement
Sustain midlife, and possibly late-life physical activity
Although we have more to learn about effectiveness, avoiding or delaying even a proportion of potentially modifiable dementias should be a national priority for all.
Interventions and care in dementia
Not all dementia will be preventable and we present the latest evidence on intervention and care for dementia. To date the emphasis has been on specific subtypes of dementia, most notably on Alzheimer's disease, which has been conceptualised over the years in a variety of changing diagnostic criteria—eg, DSM IV and DSM V. 180 , 181 Intense efforts have been put into biomarkers for early preclinical detection of the disease process before it becomes dementia. Biomarkers need to show reliability and validity, and for dementias they also need to be very closely and clearly related to clinical syndrome outcomes in the way that, for example, human papillomavirus is for cervical cancer, and hypertension has been for stroke.
Biomarkers and detection of Alzheimer's disease
Markers of neurodegeneration linked to clinical dementia include brain volume loss—ie, hippocampal volume loss and entorhinal cortex and medial temporal cortical thinning—seen in structural imaging. The most studied molecular markers are in Alzheimer's disease and are amyloid and tau, which PET and CSF detect clinically. The prevalence of particular pathologies at different ages is important in interpretation of such studies. So, for example, population derived studies show increases in plaques in the population from less than 3% at age 50–59 years to around 40% at age 80–89 years. 182
Amyloid imaging
Amyloid imaging detects amyloid in the brain with high sensitivity and specificity in both cognitively normal and people with Alzheimer's disease when the gold-standard comparison is either neuropathology or clinical diagnosis, distinguishing Alzheimer's disease from other neurodegenerative conditions. 183 Amyloid imaging is not a diagnostic test for dementia. A US study of randomly selected older people from the community recruited 1671 people (mean age of 71 years). 182 The prevalence of PET detected amyloid positivity increased from 2·7% (95% CI 0·5–4·9) of people without cognitive impairment aged 50–59 years to 41·3% (95% CI 33·4–49·2%) aged 80–89 years. 182 In 10-year follow-up PET positivity was associated with a higher probability of developing Alzheimer's disease compared with those who were amyloid negative (HR 2·6, 95% CI 1·4–4·9). In participants with mild cognitive impairment who were amyloid positive the probability (HR 1·9, 95% CI 0·9–3·9) was not very different to those who were amyloid negative (1·6, 0·8–3·4).
Similarly, an 8-year follow-up study of 599 volunteers (average age 70 years) in Australia found that cognitively normal PET amyloid-positive people had an elevated risk of developing Alzheimer's disease compared with amyloid negative (17·7% vs 8·1%; OR 2·4, 95% CI 1·5–4·0). 184 Over 80% of the 266 people who were PET amyloid-positive did not go onto develop a cognitive impairment within 8 years, showing positive status does not predict impairment for most people in a timeframe that might be a useful prognostic window. Follow-up at 5 years of amyloid-positive participants with normal cognition or mild cognitive impairment versus amyloid negative people found the same pattern of increased risk (2·6, 1·4–4·9). Risk also increases per 1 year of age (HR 1·05, 95% CI 0·55–2·0/year), and APOEε4 status (2·6, 1·4–5·0). 184
Most people who are amyloid positive with no other markers have not developed Alzheimer's disease dementia during their lifetime. A model of lifetime risks of people who are amyloid positive without any other biomarkers finds it to be 8·4% for a 90-year-old woman who is cognitively normal at baseline, 23·5% for a 75-year-old woman and 29·3% for a 65-year-old woman. 185 The 10-year risk is considerably less, so a 65-year-old woman with only amyloid biomarkers but who is cognitively normal and has no neurodegeneration has a 10-year Alzheimer's disease risk of 2·5% and a man 2·3%, but the risk is higher with accompanying neurodegeneration ( table 2 ). 185
Ten-year risks by age of developing Alzheimer's disease for women based on amyloidosis alone and in the presence of neurodegeneration and mild cognitive impairment
| 60 years | 0·2 (0·06–0·8) | 1·3 (0·6–2·5) | 3·6 (1·1–14·2) | 7·1 (4·5–10·9) | 93·5 (91·1–95·0) | 57·2 (48·2–67·9) |
| 65 years | 0·5 (0·14–1·8) | 2·5 (1·2–4·9) | 4·3 (1·4–15·0) | 10·7 (6·8–16·2) | 91·7 (89·2–93·5) | 55·4 (46·6–65·8) |
| 70 years | 1·1 (0·34–3·5) | 4·7 (2·4–8·7) | 5·5 (2·0–16·6) | 15·5 (10·0–22·8) | 88·6 (85·8–90·6) | 52·2 (43·8–62·4) |
| 75 years | 2·2 (0·74–6·5) | 7·8 (4·1–14·0) | 7·3 (2·9–19·0) | 20·8 (13·7–29·7) | 83·8 (80·7–86·2) | 47·4 (39·6–57·0) |
| 80 years | 3·7 (1·3–9·8) | 11·1 (6·0–18·7) | 9·3 (3·9–20·9) | 24·4 (16·4–33·8) | 75·8 (72·2–78·7) | 40·0 (33·1–48·6) |
| 85 years | 4·7 (1·8–11·0) | 11·5 (6·5–18·5) | 9·7 (4·3–19·3) | 23·1 (15·8–31·2) | 63·7 (59·6–67·2) | 30·0 (24·5–37·2) |
| 90 years | 3·8 (1·5–8·2) | 8·2 (4·7–12·9) | 7·1 (3·3–13·3) | 16·8 (11·5–22·6) | 46·7 (42·7–50·2) | 19·1 (15·3–24·3) |
Data are relative risk (95% CI) or %. Reproduced from Brookmeyer and Abdalla 185 by permission of Elsevier.
Overall, the knowledge of PET-measured amyloid and tau status and MRI-derived cortical thickness in a general population derived sample, only adds a small improvement, which might not be clinically important for predicting memory decline over a model with clinical and genetic variables. 186
Using amyloid PET in patients with cognitive impairment of uncertain causes, results in changes to the clinical diagnosis of Alzheimer's disease 187 and sometimes to medication prescription. We do not know whether PET use improves patient care or decreases care costs. Many people have a mixed cause of dementia and a positive result does not indicate only Alzheimer's disease.
Fluid biomarkers
PET imaging is very costly (US$3000 in the USA) and although used in some clinical settings remains the topic of research to understand its usefulness in broader populations. Fluid biomarkers—ie, blood and cerebrospinal fluid tests—have become a more practical focus of interest since it has become possible to measure specific proteins linked to the proteins associated with the neuropathologies of Alzheimer's disease. 188 A composite blood biomarker for amyloid tested in a discovery dataset and then a validation cohort of participants aged 60–90 years who were already taking part in studies in Japan or Australia had areas under the receiver operating characteristic curves of 96·7% for discovery and 94·1% for validation. The blood biomarker had sensitivity and specificity above 80% against amyloid PET measurement 188 and correlated with CSF concentrations of Aβ1–42. These results are similar to other amyloid blood biomarkers 189 , 190 and harmonisation to a common reference standard is now vital. Although CSF Aβ1–42/1–40 ratio and amyloid PET are now considered interchangeable, 191 CSF tau biomarkers have only correlated weakly with brain tau as currently measured by radioligands. 192 Neurofilament light protein is measured in many cohorts; however, it is non-specific. People with Huntington's disease, multiple sclerosis, mild cognitive impairment, and Alzheimer's disease might have raised blood neurofilament light concentrations, which are a marker of neurodegeneration. 193 , 194 , 195
Key points and conclusions
To be useful in clinical practice biomarkers must be well understood in the populations to which they are going to be applied, including the effects of age and sex on results. There is now reasonable evidence that amyloid and tau measured by PET or in fluid indicate increased risk for development of cognitive impairment in older adults but at the individual level prognostication is not possible as most cognitively normal people with these markers do not develop dementia within a clinically relevant timeframe. Negative amyloid results can be useful for ruling out current Alzheimer's pathology in people with cognitive impairment when the cause is uncertain and show an individual is unlikely to develop Alzheimer's disease during the next few years. High neurofilament light concentrations indicate a neurodegenerative process but not its cause. The value of biomarkers, in terms of diagnostic value, has not been addressed in different representative populations and particularly not in those from LMIC. The potential advantages of blood biomarkers are their low cost and their wider acceptability and applicability in many settings. In many areas of medicine more reliable diagnostic tests have improved research, including epidemiological and public health research and trials, to help distinguish cause from symptom (tuberculosis from a fever) or assess risk factor and disease (hypercholesterolaemia and ischaemic heart disease). Those biomarkers developed for the underlying biology of the dementia syndrome are subject to the same assessment of value.
Principles of intervention in people with dementia
In the 2017 Commission, we discussed that when concerns are raised by patients or family, an accurate diagnosis is helpful. Such a diagnosis provides a gateway to intervention and services where available, for planning for possible futures, and support for family, as well as to research. Unfortunately, these services are not always available. National plans for dementia support timely diagnosis and offer help to individuals and their families.
We did not address screening of those not presenting with concerns but rigorous systematic reviews by the US Task Force on Prevention have found an absence of evidence of benefit and harm. 196 The first trial of population screening took place in the USA, screening 4005 primary care patients aged 65 years or older. No clear benefit or harm in terms of quality of life, mood, or increasing diagnostic rates was found. 197 Other strategies might become more valuable in time such as sensitive awareness of risk factors, when routine records suggest an individual might be deteriorating cognitively. 198
People with dementia have complex problems with symptoms in many domains. Those providing support and any interventions must consider the person as a whole, as well as their context and their close carers, whether family or friends. Individuals' medical, cognitive, psychological, environmental, cultural, and social needs must be given consideration. 2 In the context of under provision of services, this notion is and will continue to be a challenge. Dementia, as an illness which affects cognition by definition, affects the ability to organise activities and people with dementia often need help to do what they enjoy—for example, listen to music, or go to gardens and parks. Wellbeing is one of the goals of dementia care.
Interventions once a diagnosis has been made
Cholinesterase inhibitors have a useful, modest role in improving cognition and activities of daily living in patients with mild-to-moderate Alzheimer's disease and memantine can be prescribed in combination or each drug used separately for moderate and severe Alzheimer's disease. 2 , 199 , 200 However, although available in most countries these drugs are no longer remunerated in France because it is felt that they offer only a small benefit while shifting clinician's attention from other interventions. Whether non-prescribing of this drug will help patients by removing an intervention with known benefit or be detrimental to them is unknown. 201 No advances have been reported in Aβ therapeutics, with negative results from phase 3 trials of monoclonal antibodies (eg, solanezumab, crenezumab) and inhibitors of β-secretase, a protease involved in the production of Aβ peptides. 202 Aducanumab previously abandoned as futile now has further unpublished results. Three 5HT6 antagonists and the calcium channel blocker nilvadipine 203 , 204 have also been ineffective. These drugs also show substantial impact during treatments at so-called therapeutic concentrations on the leakiness of blood vessels. The long-term impact of such side-effects is unknown. Anti-tau, anti-amyloid, and anti-inflammatory drugs continue to be in focus and some argue that pre-symptomatic interventions are necessary, especially if targeting Aβ production, but no evidence of efficacy 205 and some evidence of worsening target symptoms currently exists. 206
Cognitive training in people with dementia
A meta-analysis of 12 controlled trials of 389 people with mild dementia, completing 4 or more hours of group-based computerised cognitive training (mean age 66–81 years, 63·5% female participants), found a small, statistically significant beneficial effect on overall cognition, driven by two trials of virtual reality or Video games (SMD=0·3, 95% CI 0·0–0·5), one with a low and one with a high risk of bias. 55
A Cochrane review 207 found 33 trials of cognitive training, only one of which overlapped with the study above, with around 2000 participants with mild-to-moderate dementia, most with a high or uncertain risk of bias. 207 People completing cognitive training, compared with usual treatment or non-specific activities, had small-to-moderate effects on overall cognition (SMD 0·4, 95% CI 0·2–0·6) and specific cognitive abilities such as verbal fluency and improvements lasted for a few months to 1 year. No direct evidence was observed to suggest that cognitive training was better than cognitive stimulation therapy.
Exercise and physical activity
The Dementia and Physical Activity RCT 208 found moderate-to-high intensity aerobic and strength exercise training did not slow cognitive impairment in people with mild-to-moderate dementia but improved physical fitness. The US Reducing Disability in Dementia study 209 implemented an at-home multicomponent intervention including exercise education, training to increase pleasant events, and activator-behaviour-consequence problem-solving approach over 6 weeks by case managers in 255 community dwelling people with dementia older than 60 years and their family carer and were able to follow up 140 (54·9%). The study found increased physical activity; days of taking 30 or more minutes of exercise (effect size 0·6, 95% CI 0·4–0·8 after the treatment and 0·3, 0·1–0·5 at 13 months) in a before and after intervention comparison.
Interventions for neuropsychiatric symptoms of dementia
Neuropsychiatric symptoms are common and often clustered in people with dementia. These symptoms might precede dementia and are associated with tau and amyloid neuropathology. 210 This suggests that underlying neurobiological mechanisms might underpin neuropsychiatric symptoms. However, other drivers relating to the personal history and the environment of the person with dementia are also likely to exist. Neurodegeneration could lead to increased vulnerability to stressors or triggers. Genetics, cognitive reserve, resilience, medical comorbidities, and environment including responses of carers might modify these relationships. Needs and responses will also be individual and relate to a person's own social, cultural, and historical context. First-line assessment and management of neuropsychiatric symptoms should focus on basic health: describe and diagnose symptoms; look for causes such as pain (using validated pain assessments might help), illness, discomfort, hunger, loneliness, boredom, lack of intimacy and worry that could cause the behaviours and alleviate these while considering risks of harm. 2
No new evidence of medication effectiveness for these symptoms exists; risperidone in low doses (0·5 mg daily) and some other antipsychotics are sometimes effective but often ineffective and have adverse effects. 2 Specific initiatives have led to a decrease in antipsychotic prescriptions for people with dementia, although often replaced with other psychotropics ( figure 8 ), such as benzodiazepines, antidepressants, and mood stabilisers. 211 These psychotropics lack evidence of efficacy for neuropsychiatric symptoms but show clear evidence of possible harm; for example, trazodone and benzodiazepines increase fall-related injuries. 144 Major policy changes should be assessed carefully, within and across countries for unintended consequences (and perhaps unexpected benefits) and their costs.
Proportion of patients with a diagnosis of dementia prescribed an antipsychotic drug (A) and those prescribed an anxiolytic, hypnotic, or antidepressant (B)
CPRD=Clinical Practice Research Datalink. Reproduced from Donegan et al, 211 by permission of Elsevier.
Evidence is slowly accumulating for the effectiveness, at least in the short term, of person-centred evidence-based psychosocial interventions. In Germany, a 6-month cluster RCT of nurse-delivered, supervised dementia care management used a computer-assisted nurse assessment to determine personalised intervention modules, then a multi-disciplinary team discussion and agreement with the physician for 634 people (mean age 80 years) with dementia living at home with a primary carer or alone. 212 The mean mini mental state examination (MMSE) was 23, only 38% had a formal diagnosis of dementia; the majority of participants (51%) had mild dementia but some had moderate and some severe dementia. The intervention consisted of psychosocial management of treatment and care, medication management and carer support, and education and discussion with a psychiatrist or neurologist. The intervention, compared with care as usual, was associated with better outcomes for neuropsychiatric symptoms (Neuropsychiatric Inventory [NPI] score −7·5, 95% CI −11·1 to −3·8), however this effect could be because of deterioration in care as usual (in the care as usual group NPI increased from 7·2 to 15·2; in the intervention group NPI increased from 7·6 to 8·2). This between-group reduction in neuropsychiatric symptoms was greater than that expected, extrapolating from other study results, with antipsychotic medication. Effects on quality of life were only apparent for those people living with a carer.
An eight-session home-based tailored activity programme RCT, tailored both to the person with dementia living at home and to a family member compared with eight telephone-based education sessions, recruited 160 participants with 64% follow-up, imputing values for the rest. 213 The study reported a large reduction in overall neuropsychiatric symptoms immediately after the intervention, which were better in the group receiving home-based tailored activity programme on the neuropsychiatric inventory (mean difference in score 24·3, 95% CI 3·1–45·6), and on functional dependence and pain but this was not sustained 4 months later. Non-completers had more severe neuropsychiatric symptoms.
Since the 2017 Commission two new systematic reviews of antidepressants to treat depression in dementia reported moderate quality evidence that antidepressant treatment for people with dementia does not lead to better control of symptomatology compared with placebo. 214 , 215
Agitation is distressing for people with dementia and those around them, and contributes substantially to the overall costs as the level of agitation increases. 216 The body of evidence on this key behaviour is growing, mostly focused on care-home settings. These findings are valuable as these populations are most affected; however, because many people with dementia reside at home a major gap in knowledge remains.
Care home residents with agitation often find sitting still difficult and therefore might not be included in activities. 217 , 218 Two new cluster RCTs of professionals delivering multicomponent, interdisciplinary, interventions in care homes successfully reduced agitation. The WHELD study 219 included participants with or without neuropsychiatric symptoms and provided person-centred care, aiming to improve communication with people with dementia. It implemented social, sensory experiences or other activities; educated about antipsychotic review; and addressed physical problems, finding lower Cohen Mansfield Agitation Inventory (CMAI) at 9 months (MD −4·3 points, 95% CI −7·3 to −1·2). 219 The TIME study 220 for people with moderate-to-high levels of agitation consisted of a manual-based comprehensive assessment of the resident and structured case conference for the staff and doctor, to create a tailored plan, and then implement it. This intervention led to reduced agitation at 8 weeks (NPI −1·1 points, 95% CI −0·1 to −2·1; CMAI −4·7 points, −0·6 to −8·8) and 12 weeks (NPI −1·6, −0·6 to −2·7; CMAI −5·9, −1·7 to −10·1). 220 These effect sizes are similar to those seen for medications, but without harmful side-effects. 2 , 221 A further RCT studied a six-session intervention with staff in groups, teaching staff to understand agitation as related to medical, psychological, or social unmet needs and to implement strategies to meet these needs, using the describe, investigate, create, and evaluate approach. 222 The intervention did not reduce agitation symptoms, although it was cost-effective, improving quality of life. 223 Overall, the current evidence for agitation in care homes favours multi-component interventions by clinical staff, including considering if drugs might harm, and not drug interventions. Thus a major gap remains in knowledge about people living at home who comprise the majority of those with dementia.
Psychotic symptoms in dementia
People with dementia might be wrongly thought to have delusions when they misremember, and new psychotic symptoms are often due to delirium, thus thorough assessment of symptoms is essential. 2 Management of psychosis in dementia should start with non-pharmacological interventions; however, evidence for effectiveness of these interventions for psychosis in dementia is weaker than for agitation. 224 Antipsychotics for psychosis in dementia should be prescribed in as low a dose and for the shortest duration possible. 2 However, a Cochrane review of antipsychotics withdrawal found two trials with participants with dementia who had responded to antipsychotic treatment. These reported that stopping antipsychotics was associated with symptomatic relapse 225 suggesting the need for caution in any medication withdrawal in this group. There was low-quality evidence that, in general, discontinuation might make little or no difference to overall neuropsychiatric symptoms, adverse events, quality of life or cognitive function. 226
Apathy might be conceptualised as the opposite of engagement, comprising reduced interest, initiative, and activity. Like people without dementia, those with dementia engage more in preferred activities, but require additional support to do so. 227 A study in care homes observed engagement increased during activities in those who attended the groups. 228 A Cochrane review of the few people who had been in drug RCTs of methylphenidate versus placebo for apathy in dementia found small improvements on the apathy evaluation scale (MD −5·0, 95% CI −9·6 to−0·4, n=145, three studies, low-quality evidence) but not on the NPI apathy subscale (MD −0·1, 95% CI −3·9 to 3·7, n=85, two studies). 229
There is no evidence that medication for sleep in dementia is effective 230 and considerable evidence for harm—ie, earlier death, increased hospitalisation, and falls—exists. 139 , 144 Testing of non-pharmacological interventions is ongoing. 231
Carer distress related to neuropsychiatric symptoms rather than the dementia symptoms was associated in one study with increased use and costs of health services, 232 highlighting the need for effectively identifying, educating, and supporting distressed carers. An RCT 233 reporting 6-year follow-up after the eight session STrAtegies for RelaTives intervention—manual-based coping intervention delivered by supervised psychology graduates—found continuing effectiveness for depressive symptoms in carers (adjusted MD −2·00; 95% CI −3·4 to −0·6) and risk of case-level depression, with patient-related cost being approximately 3 times lower than those who did not receive the intervention (median £5759 vs £16 964 in the final year; p=0·07). 233 Another US study 234 followed up 663 people, mean age 77 years, 55% women. Caregiver depression rather than symptoms of people with dementia predicted emergency department use for people with dementia, with a 73% (RR 1·73, 95% CI 1·3–2·3) increase. 234
Functioning
A UK RCT of 14 sessions of cognitive rehabilitation focused on individual goal attainment with therapy delivered at home by an occupational therapist or nurse to 475 participants with mild-to-moderate dementia (MMSE ≥18 for inclusion; mean 24) and a family carer. 235 Individuals had two or three goals; the most common was engaging in activities (21% of goals). The intervention group reported increased goal attainment over 3 and 9 months compared with usual treatment (effect size 0·8, 95% CI 0·6–1·0 at both 3 and 9 months). 235 The treatment did not improve participants' quality of life, mood, self-efficacy, cognition, carer stress, or health status and was not cost-effective. A systematic review 236 of RCTs without meta-analysis for overall effect size, concluded that all interventions which had improved functioning in people living with dementia in the community have been individual rather than group interventions. These were: in-home physiotherapist delivered aerobic exercise (two studies, larger one positive, 140 people with Alzheimer's disease; smaller study negative, 30 people with Alzheimer's disease), individualised cognitive rehabilitation (mild or moderate dementia; two studies; 257 cognitive reserve intervention groups and 255 controls), and in-home activities-focused occupational therapy (people with mild to moderate dementia, three studies, 201 intervention, 191 controls) reduced functional decline compared to controls but group-exercise and reminiscence therapies were ineffective. 236
People with dementia have other illnesses
Multimorbidity is a huge challenge in dementia, not only because people with dementia have increased rates of other illnesses, but also because they often find it particularly difficult to organise care. People with dementia might forget to tell their family or health professionals of symptoms, struggle to understand or follow agreed plans, and are more likely to forget to drink and eat, increasing falling and infection rates. 237 People with dementia consult primary care less often 238 and have fewer dental visits 239 than those without dementia and their family members, if involved, often feel they lack knowledge to assist. 240 Health-care professionals need education to be more comfortable, understanding, and positive in communicating with people with dementia. 241
Around 70–80% of people diagnosed with dementia in primary care have at least two other chronic illnesses. 242 , 243 People who are physically more frail are more likely to have dementia, but the relationship between pathology and symptoms in these people is comparatively weak suggesting that dementia might be from other causes. 22 Compared to the general older population, people with dementia have increased rates of cerebrovascular disease, 243 , 244 , 245 , 246 stroke, 247 Parkinson's disease, 243 , 245 diabetes, 245 , 247 skin ulcers, anxiety and depression, 243 , 245 pneumonia, incontinence, and electrolyte disturbance. 245 Multimorbidity in people with dementia is associated with faster functional decline 248 and worse quality of life for people with dementia and their family carers. 249
Dementia and COVID-19
Severe acute respiratory syndrome coronavirus 2, was first identified in patients with viral pneumonia in Hubei province, China. 250 Severity and mortality of the associated disease (COVID-19) worsen with increasing age 251 and with pre-existing illnesses such as hypertension and diabetes, 252 and thus many people with dementia are at particular risk. Death certificates from the UK indicate that dementia and Alzheimer's disease were the most common underlying conditions, specified in 11 950 deaths (25·6% of all deaths involving COVID-19) in March to May, 2020. 253 Many charities, practitioners, and academics supporting people with dementia have issued guidance based on current evidence and best practice, including advance consideration of whether people would wish to be hospitalised if they develop severe COVID-19. Concern has been expressed that the illness and consequent distancing might increase family carer stress, loneliness, neuropsychiatric symptoms and use of psychotropic medication, and lead to complications, including future dementia. Interventions delivered remotely through technology have also been implemented in some places. 254 , 255 , 256 , 257
People with dementia might struggle to adhere to measures to reduce virus transmission, as they might not understand or remember about required changes to behaviour, such as physical distancing and hygiene, leading to increased risk to themselves and their carers. 258 They might additionally be vulnerable if they depend on others for daily activities or personal care, as this necessitates close personal contact.
This situation is particularly concerning in those care homes, where many residents have dementia and where many COVID-19 deaths have occurred in many countries 259 , 260 , 261 with reports of more than half of residents being admitted to hospital. In US nursing homes, among 10 576 people with confirmed COVID-19, residents living with dementia made up 52% of COVID-19 cases; yet, accounted for 72% of all deaths (an increased risk of 1·7). 262 The number of people living together in care homes means that the infection of an individual, either staff or resident, could endanger more people than in traditional or family households. Although evidence exists that if staff are sufficiently and rigorously protected they are unlikely to develop COVID-19, many staff have become unwell and some have died. 263 , 264 Illness means that there are fewer people to care for residents at a time when they need particularly high levels of care. This situation is particularly relevant in the care of residents with dementia, if they are expected to remain in their own rooms, rather than eating and participating in activities with others. Staff or residents might also be moved between care homes and increase risk in other homes. 261 Restrictions on visitors to private homes, care homes, and hospitals might cause greater distress for people with dementia and they might not understand why people are wearing masks, recognise who is behind it, or understand speech when lips are covered. Lack of restrictions means that the visitors might also be at elevated risk. 261
The impacts of COVID-19 on people with dementia might be particularly severe in LMICs, due to smaller health budgets for testing and protective equipment, capacity of health-care systems, quality of care home provision and patterns of workforce mobility. 264
Thus, people with dementia are particularly vulnerable to COVID-19 because of their age, multimorbidity, and difficulties in maintaining physical distancing. 250 , 251 , 252
We recommend rigorous public health measures of protective equipment and hygiene, including not moving staff or residents between care homes or admitting new residents when their COVID-19 status is unknown, should mitigate impacts on people with dementia. It is also imperative that there is frequent and regular testing of staff in care homes for infection, ensuring staff have sick pay so that they do not come in when symptomatic and interim care is being set up for people discharged from hospital so that only those who are COVID-19 free come to live in care homes. Resident testing should encompass asymptomatic as well as symptomatic people, when there is exposure within the home to COVID-19. In the future, many homes might be able to start to provide oxygen therapy so that those who do not want to be admitted to hospital are still able to access oxygen therapy. In addition, it is also important to reduce isolation by providing the necessary equipment and a brief training to relatives on how to protect themselves and others from COVID-19; so that they can visit their relatives with dementia in nursing homes safely when it is allowed. Further evidence is needed to inform responses to this and future public health emergencies.
Hospital admissions
Hospitalisation in people with dementia is associated with adverse, unintended consequences, including distress, functional and cognitive decline, and high economic costs. 265 , 266 , 267 People with dementia have 1·4 to 4 times more hospital admissions than others with similar illnesses. 266 , 268 , 269 , 270
A systematic review and meta-analysis including 34 studies of 277 432 people with dementia found that in the six studies which compared the two groups, people with dementia had increased hospital admissions compared with those without dementia, after adjusting for age, sex, and physical comorbidity (RR 1·4, 95% CI 1·2–1·7; figure 9 ). 271 Hospitalisation rates in people with dementia ranged from 0·37 to 1·26 per person-year in high-quality studies. Admissions are often for conditions that might be manageable in the community (potentially preventable hospitalisations). 268 People with dementia experience longer and more frequent admissions and readmissions; health-care expenditure for people with moderate-severe dementia is around double that of people without dementia. 269 , 272 , 273 Early detection and management of physical ill-health in people with dementia, particularly of pain, falls, diabetes, incontinence, and sensory impairment, is important. 199 , 274 , 275 However, no intervention has successfully reduced number of hospital admissions of community-dwelling people with dementia, 276 although education, exercise, rehabilitation, and telemedicine have reduced admissions for older people without dementia. 277
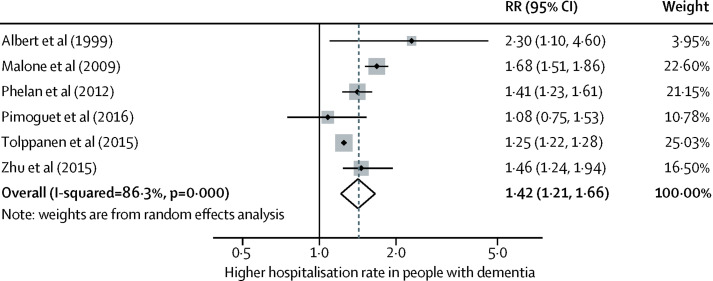
Systematic review and meta-analysis of hospitalisation rates of people with dementia compared to those without dementia controlled for age and sex
Reproduced from Shepherd et al, 271 by permission of Springer Nature.
High-quality care for people with dementia takes longer than caring for others with the same condition. 278 Recognition of dementia in hospital inpatients is necessary for optimum care, 279 but dementia is often undetected or unrecorded. 280 In the UK however, detection rates have increased over the past 10 years. 281
Physical illness, delirium, and dementia
Dementia and delirium frequently occur together. In one hospital inpatients' survey nearly 35% of those older than 80 years experienced delirium; those with prior cognitive impairment had 15 times the risk of developing delirium than those without (OR 15·3, 95% CI 5·2–45·4). 282 People with delirium without known dementia are more likely to be diagnosed with dementia in the future than others, either because of pre-existing undiagnosed dementia or cognitive impairment, present in 20·7% (95% CI 11·9–29·5) and 37·8% (27·3–88·3) respectively of one cohort, or because delirium has neurotoxic effects and so precipitates dementia. 283 People with similar neuropathology show faster cognitive decline if they develop delirium than if they do not. 284 Additionally, older people without dementia declined cognitively more than twice as fast after an emergency hospital admission for any cause, compared with those not admitted, suggesting any severe illness is associated with cognitive decline. 285 Risk factors for delirium in dementia include sensory impairment, pain, polypharmacy, dehydration, intercurrent illnesses, such as urinary tract infections or faecal impaction, and an unfamiliar or changing environment. 286 Delirium in older people should prompt consideration of underlying dementia.
Most research on delirium prevention has been in people without dementia. It suggests targeting hydration, stopping medication predisposing to delirium, monitoring the depth of anaesthesia, and sleep promotion. However, no evidence for medication efficacy, including cholinesterase inhibitors, antipsychotic medication, or melatonin exists. 287 , 288 , 289 The Hospital Elder Life Program 290 —an intervention to prevent delirium in those admitted to hospital—reduces delirium incidence and includes people who are cognitively impaired. This multidisciplinary treatment consists of daily visits, orientation, therapeutic activities, sleep enhancement, early mobilisation, vision and hearing adaptation, fluid repletion, infection prevention and management of constipation, pain, and hypoxia, and feeding assistance. 290
A network meta-analysis of drugs for prevention and treatment of delirium did not include studies of people with dementia, thus we cannot use this to recommend drugs for people with dementia and delirium as this research might be inapplicable to them. 291
Little high-quality research exists on managing delirium in dementia. One RCT compared care at a specialist medical and mental health unit to usual care for 600 confused people older than 65 years, acutely admitted to hospital and found no difference in the primary outcome of days spent at home or in hospital, but increased family satisfaction. 292 A further RCT of cognitively stimulating activities for people with delirium in dementia did not improve the delirium. 293 No definitive evidence that any medication improves delirium in people with dementia exists: cholinesterase inhibitors, antipsychotics, and sedating benzodiazepines are ineffective and antipsychotics and benzodiazepines are associated with mortality and morbidity. 265 , 288 , 294 , 295 , 296 , 297 Given the risk of dementia in people who develop delirium, its prevention, and possibly advances in its management, might offer a means for dementia prevention. 298
Link between very old age, frailty, and dementia
The fastest growing demographic group in most advanced countries are people aged 90 years and older. One well characterised post-mortem cohort of the oldest old (n=1079; mean age 90 years) dying with dementia, found that neuropathological features of Alzheimer's disease account for about half of the cognitive decline seen as people diagnosed with Alzheimer's disease had mixed causes of dementia. 299 Although Alzheimer's disease neuropathology was the commonest cause of dementia, Alzheimer's disease changes rarely occurred on their own, so only 9% of people with dementia had pure Alzheimer's disease pathology. 300 People who have Alzheimer's disease pathology without developing dementia tend to have fewer age-related health deficits than those who develop it with even low concentrations of plaques and tangles. 301 A moderation analysis showed that the relationship between Alzheimer's disease pathology and dementia status differed according to level of frailty (adjusted for age, sex, and education) with increasing frailty weakening the relationship between Alzheimer's disease pathology and dementia ( figure 10 ). 22 As with delirium, some of this additional health risk might be modifiable. This approach suggests a new type of therapy focus on specific age-related processes that underpin many diseases of late life might reduce the incidence or severity of dementia.
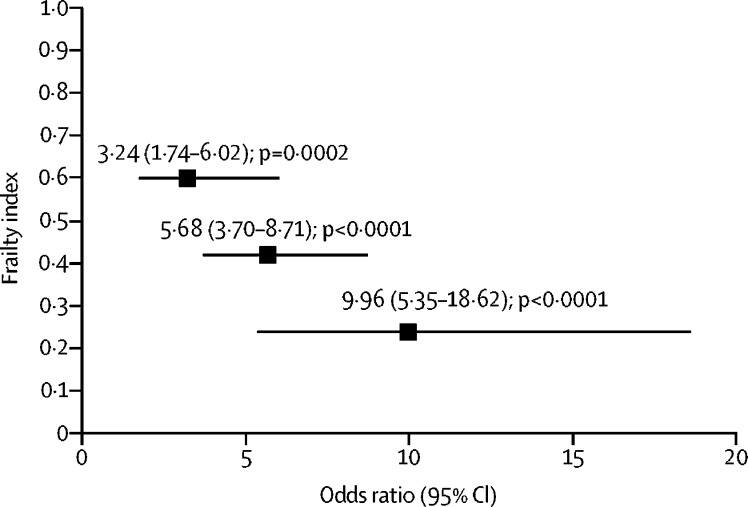
Moderation analyses of the relationship between Alzheimer's disease pathology and clinical diagnosis of Alzheimer's dementia (adjusted for age, sex, and education)
As frailty increased, the odds of a neuropathological diagnosis of Alzheimer disease corresponding to a clinical diagnosis decreased. Reproduced from Wallece et al, 22 by permission of Elsevier.
End-of-life care in dementia
The numbers of people dying with dementia are increasing but the evidence for the best end-of-life care is scarce. Trends in age-standardised death rates (3·6%) for dementia increased slightly between 1990–2016, with pronounced increases in the USA and Japan and decreases in western Europe and central Latin America. 4 Dementia is more readily being included on death certificates, which accounts for some of the rise. The increase might be related to dementia manifesting at later ages, with higher physical frailty 22 leading to a faster decline.
Most people with dementia might die while still in the mild-to-moderate stages whereas only about a quarter of those dying with dementia have severe dementia. 302 , 303 The trajectory of dementia is often unpredictable 304 and palliative care initiation should reflect need not prognosis.
Decision making about end of life is complex and simple rules of thumb, co-designed with staff and carers, provided clarity in some small studies. 304 One RCT testing decision-aids about families' and doctors' goals of care for people with advanced dementia led to increased palliative care content in care plans. 305 , 306 In a 9-month UK prospective study, 85 care home residents with advanced dementia from 14 homes were likely to be living with distressing symptoms, specifically agitation (54%) or pain (61% on movement). 304
Capacity to make abstract decisions, including about the future, might be lost early in dementia. 307 Therefore, advance care planning, designed to empower people with dementia and improve quality of dying, might theoretically be something everyone should do before developing dementia. 308 However, people might not be able to predict their future wishes. This might explain why family carer proxies show only low-to-moderate agreement with stated end-of-life treatment preferences of people with dementia. 309 Advance care planning might, however, reduce carers' uncertainty in decision making and improve perceptions of quality of care. 310
Partners of people dying with dementia experience poorer mental health than those facing bereavement from other causes 311 possibly because of long and difficult caring responsibilities. This might be ameliorated through sensitive and timely information, particularly regarding the progression of dementia, 312 individually or through family and staff case-conferencing. 313 , 314
Conclusions
Knowledge about risk factors and potential prevention, detection, and diagnosis of dementia is improving although significant gaps remain. 315 In this Commission report, we have specified policy and individual changes to delay the onset of cognitive impairment and dementia and better ways to support and treat people with dementia and their families and to improve their quality of life.
Interventions, including organisation of the complex physical illness and social needs, to support people affected by dementia can have a huge effect when taken as a whole. Our ambition is for worldwide provision of resources for an adequate level of wellbeing to people with dementia and their carers with a better evidence base to guide individual care and policy making alike. With good quality care, people can live well with dementia and families can feel supported.
Acknowledgments
We are partnered by University College London (UCL), the Alzheimer's Society, UK, the Economic and Social Research Council, and Alzheimer's Research UK, and would like to thank them for financial help. These organisations funded the fares, accommodation, and food for the Commission meeting but had no role in the writing of the manuscript or the decision to submit it for publication. We would like to thank Bernadette Courtney, Jacques Gianino, and Nuj Monowari, from UCL, London, UK, for their administrative help, including managing finances, booking rooms and food, and setting up a website supported by the University College London Hospitals National Institute for Health Research Biomedical Research Centre. We would like thank Henrik Zetterberg for advice on biomarkers and dementia.
Contributors
GL, JH, AS, and NM contributed to literature searches and quality assessments for systematic reviews. JH and NM performed meta-analyses. GL, JH, AS, and NM conceived the new PAF calculation and NM led the statistical analysis. GL, JH, AS, NM, DA, CLB, SB, AB, JC-M, CC, SGC, NF, RH, HCK, EBL, VO, KRi, KRo, ELS, QS, LSS, and GS attended the conference to discuss the content. GL, JH, EBL, AS, DA, and ELS wrote first drafts of sections of the paper. GL wrote the first draft of the whole paper and revisions of drafts. CBa reviewed and contributed to revision of the final drafts. All authors contributed to sections of the reports and all revised the paper for important intellectual content.
Declaration of interests
AS reports grants from Wellcome Trust (200163/Z/15/Z), outside the submitted work. DA reports grants from Eli Lilly, during the conduct of the study. CBa reports grants and personal fees from Aca-dia and Lundbeck; and personal fees from Roche, Otsuka, Biogen, Eli Lilly, and Pfizer, outside the sub-mitted work. SB reports grants and personal fees from AbbVie, personal fees and non-financial sup-port from Eli Lilly, and personal fees from Eleusis, Daval International, Boehringer Ingelheim, Axovant Sciences, Lundbeck, and Nutricia, outside the submitted work; and he has been employed by the Department of Health for England. NF reports non-financial support from Eli Lilly, outside the submitted work. LNG and her institutions (Johns Hopkins University, Baltimore, MD, USA, Drexel University, Philadelphia, PA, USA, and Thomas Jefferson University, Philadelphia, PA, USA) are entitled to receive royalties from fees associated with online training for the tailored activity program, which is an evidence-based program referenced in the Review. RH reports grants from Department of Health, NIHR HTA Programme, outside the submitted work; and he is a Scientific Trustee of the charity Alzheimer's Research UK. MK reports grants from the UK Medical Research Council (S011676, R024227), NordForsk (the Nordic Programme on Health and Welfare, 75021) and the Academy of Finland (311492), outside the submitted work. EBL reports other (royalties) from UpToDate, outside the submitted work. KRo reports personal fees from Clinical Cardio Day-Cape Breton University, Sydney, NS, Canada, CRUIGM-Montreal, Jackson Laboratory, Bar Harbor, MA, USA (speaker fees), MouseAge, Rome, Italy (speaker fees), Lundbeck, Frontemporal Dementia Study-Group, SunLife Insurance, Japan, outside the submitted work. He is a President and Chief Science Officer of DGI Clinical, which in the last 5 years has contracts with pharma and device manufacturers (Baxter, Baxalta, Shire, Hollister, Nutricia, Roche, Otsuka) on individualised outcome measurement. In 2017, he attended an advisory board meeting with Lundbeck. He is also Associate Director of the Canadian Consortium on Neurodegeneration in Aging, which is funded by the Canadian Institutes of Health Research, and with additional funding from the Alzheimer Society of Canada and several other charities, as well as, in its first phase (2013-2018), from Pfizer Canada and Sanofi Canada. He receives career support from the Dalhousie Medical Research Foundation as the Kathryn Allen Weldon Professor of Alzheimer Research, and research support from the Canadian Institutes of Health Research, the QEII Health Science Centre Foundation, the Capital Health Research Fund and the Fountain Family Innovation Fund of the QEII Health Science Centre Foundation. LSS reports grants and personal fees from Eli Lilly, Merck, and Roche/Genentech; personal fees from Avraham, Boehringer Ingelheim, Neurim, Neuronix, Cognition, Eisai, Takeda, vTv, and Abbott; and grants from Biogen, Novartis, Biohaven, and Washington University DIAN-TU, outside the submitted work. The remaining authors declare no conflict of interests.

Supplementary Material
- 1. Patterson C. Alzheimer's Disease International; London: 2018. World Alzheimer report 2018. [ Google Scholar ]
- 2. Livingston G, Sommerlad A, Orgeta V, et al. Dementia prevention, intervention, and care. Lancet. 2017;390:2673–2734. doi: 10.1016/S0140-6736(17)31363-6. [ DOI ] [ PubMed ] [ Google Scholar ]
- 3. Nelson PT, Dickson DW, Trojanowski JQ, et al. Limbic-predominant age-related TDP-43 encephalopathy (LATE): consensus working group report. Brain. 2019;142:1503–1527. doi: 10.1093/brain/awz099. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 4. Collaborators GBDD. Global, regional, and national burden of Alzheimer's disease and other dementias, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019;18:88–106. doi: 10.1016/S1474-4422(18)30403-4. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 5. Wu Y-T, Beiser AS, Breteler MMB, et al. The changing prevalence and incidence of dementia over time - current evidence. Nat Rev Neurol. 2017;13:327–339. doi: 10.1038/nrneurol.2017.63. [ DOI ] [ PubMed ] [ Google Scholar ]
- 6. Kingston A, Comas-Herrera A, Jagger C. Forecasting the care needs of the older population in England over the next 20 years: estimates from the Population Ageing and Care Simulation (PACSim) modelling study. Lancet Public Health. 2018;3:e447–e455. doi: 10.1016/S2468-2667(18)30118-X. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 7. Gao S, Burney HN, Callahan CM, Purnell CE, Hendrie HC. Incidence of dementia and alzheimer disease over time: a meta-analysis. J Am Geriatr Soc. 2019;67:1361–1369. doi: 10.1111/jgs.16027. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 8. Friedrich MJ. Global obesity epidemic worsening. JAMA. 2017;318:603. doi: 10.1001/jama.2017.10693. [ DOI ] [ PubMed ] [ Google Scholar ]
- 9. Singh-Manoux A, Dugravot A, Shipley M, et al. Obesity trajectories and risk of dementia: 28 years of follow-up in the Whitehall II study. Alzheimers Dement. 2018;14:178–186. doi: 10.1016/j.jalz.2017.06.2637. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 10. Ahmadi-Abhari S, Guzman-Castillo M, Bandosz P, et al. Temporal trend in dementia incidence since 2002 and projections for prevalence in England and Wales to 2040: modelling study. BMJ. 2017;358 doi: 10.1136/bmj.j2856. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 11. Kivimäki M, Luukkonen R, Batty GD, et al. Body mass index and risk of dementia: analysis of individual-level data from 1·3 million individuals. Alzheimers Dement. 2018;14:601–609. doi: 10.1016/j.jalz.2017.09.016. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 12. McGrath ER, Beiser AS, DeCarli C, et al. Blood pressure from mid- to late life and risk of incident dementia. Neurology. 2017;89:2447–2454. doi: 10.1212/WNL.0000000000004741. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 13. Abell JG, Kivimäki M, Dugravot A, et al. Association between systolic blood pressure and dementia in the Whitehall II cohort study: role of age, duration, and threshold used to define hypertension. Eur Heart J. 2018;39:3119–3125. doi: 10.1093/eurheartj/ehy288. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 14. Delgado J, Bowman K, Ble A, et al. Blood pressure trajectories in the 20 years before death. JAMA Intern Med. 2018;178:93–99. doi: 10.1001/jamainternmed.2017.7023. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 15. Stern Y, Arenaza-Urquijo EM, Bartrés-Faz D, et al. Whitepaper: defining and investigating cognitive reserve, brain reserve, and brain maintenance. Alzheimers Dement. 2020 doi: 10.1016/j.jalz.2018.07.219. published online Jan 6. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 16. Perneczky R, Kempermann G, Korczyn AD, et al. Translational research on reserve against neurodegenerative disease: consensus report of the International Conference on Cognitive Reserve in the Dementias and the Alzheimer's Association Reserve, Resilience and Protective Factors Professional Interest Area working groups. BMC Med. 2019;17:47. doi: 10.1186/s12916-019-1283-z. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 17. Cholerton B, Larson EB, Baker LD, et al. Neuropathologic correlates of cognition in a population-based sample. J Alzheimers Dis. 2013;36:699–709. doi: 10.3233/JAD-130281. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 18. Latimer CS, Keene CD, Flanagan ME, et al. Resistance to Alzheimer disease neuropathologic changes and apparent cognitive resilience in the Nun and Honolulu-Asia Aging Studies. J Neuropathol Exp Neurol. 2017;76:458–466. doi: 10.1093/jnen/nlx030. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 19. Arenaza-Urquijo EM, Przybelski SA, Lesnick TL, et al. The metabolic brain signature of cognitive resilience in the 80+: beyond Alzheimer pathologies. Brain. 2019;142:1134–1147. doi: 10.1093/brain/awz037. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 20. Franzmeier N, Düzel E, Jessen F, et al. Left frontal hub connectivity delays cognitive impairment in autosomal-dominant and sporadic Alzheimer's disease. Brain. 2018;141:1186–1200. doi: 10.1093/brain/awy008. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 21. Neitzel J, Franzmeier N, Rubinski A, Ewers M. Left frontal connectivity attenuates the adverse effect of entorhinal tau pathology on memory. Neurology. 2019;93:e347–e357. doi: 10.1212/WNL.0000000000007822. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 22. Wallace LMK, Theou O, Godin J, Andrew MK, Bennett DA, Rockwood K. Investigation of frailty as a moderator of the relationship between neuropathology and dementia in Alzheimer's disease: a cross-sectional analysis of data from the Rush Memory and Aging Project. Lancet Neurol. 2019;18:177–184. doi: 10.1016/S1474-4422(18)30371-5. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 23. van der Lee SJ, Teunissen CE, Pool R, et al. Circulating metabolites and general cognitive ability and dementia: evidence from 11 cohort studies. Alzheimers Dement. 2018;14:707–722. doi: 10.1016/j.jalz.2017.11.012. [ DOI ] [ PubMed ] [ Google Scholar ]
- 24. Peters R, Ee N, Peters J, Booth A, Mudway I, Anstey KJ. Air pollution and dementia: a systematic review. J Alzheimers Dis. 2019;70:S145–S163. doi: 10.3233/JAD-180631. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 25. Chieffi S, Messina G, Villano I, et al. exercise influence on hippocampal function: possible involvement of orexin-A. Front Physiol. 2017;8:85. doi: 10.3389/fphys.2017.00085. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 26. Parbo P, Ismail R, Hansen KV, et al. Brain inflammation accompanies amyloid in the majority of mild cognitive impairment cases due to Alzheimer's disease. Brain. 2017;140:2002–2011. doi: 10.1093/brain/awx120. [ DOI ] [ PubMed ] [ Google Scholar ]
- 27. Anstey KJ, Ee N, Eramudugolla R, Jagger C, Peters R. A systematic review of meta-analyses that evaluate risk factors for dementia to evaluate the quantity, quality, and global representativeness of evidence. J Alzheimers Dis. 2019;70:S165–S186. doi: 10.3233/JAD-190181. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 28. Prince M, Ferri CP, Acosta D, et al. The protocols for the 10/66 dementia research group population-based research programme. BMC Public Health. 2007;7:165. doi: 10.1186/1471-2458-7-165. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 29. Mukadam N, Sommerlad A, Huntley J, Livingston G. Population attributable fractions for risk factors for dementia in low-income and middle-income countries: an analysis using cross-sectional survey data. Lancet Glob Health. 2019;7:e596–e603. doi: 10.1016/S2214-109X(19)30074-9. [ DOI ] [ PubMed ] [ Google Scholar ]
- 30. Wu YT, Ali GC, Guerchet M, et al. Prevalence of dementia in mainland China, Hong Kong and Taiwan: an updated systematic review and meta-analysis. Int J Epidemiol. 2018;47:709–719. doi: 10.1093/ije/dyy007. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 31. Hoffman SJ, Mammone J, Rogers Van Katwyk S, et al. Cigarette consumption estimates for 71 countries from 1970 to 2015: systematic collection of comparable data to facilitate quasi-experimental evaluations of national and global tobacco control interventions. BMJ. 2019;365 doi: 10.1136/bmj.l2231. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 32. Jia L, Quan M, Fu Y, et al. Dementia in China: epidemiology, clinical management, and research advances. Lancet Neurol. 2019;19:81–92. doi: 10.1016/S1474-4422(19)30290-X. [ DOI ] [ PubMed ] [ Google Scholar ]
- 33. Sabia S, Dugravot A, Dartigues JF, et al. Physical activity, cognitive decline, and risk of dementia: 28 year follow-up of Whitehall II cohort study. BMJ. 2017;357 doi: 10.1136/bmj.j2709. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 34. Singh-Manoux A, Dugravot A, Fournier A, et al. Trajectories of depressive symptoms before diagnosis of dementia: a 28-year follow-up study. JAMA Psychiatry. 2017;74:712–718. doi: 10.1001/jamapsychiatry.2017.0660. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 35. Norton S, Matthews FE, Barnes DE, Yaffe K, Brayne C. Potential for primary prevention of Alzheimer's disease: an analysis of population-based data. Lancet Neurol. 2014;13:788–794. doi: 10.1016/S1474-4422(14)70136-X. [ DOI ] [ PubMed ] [ Google Scholar ]
- 36. Satizabal CL, Beiser AS, Chouraki V, Chêne G, Dufouil C, Seshadri S. Incidence of dementia over three decades in the Framingham heart study. N Engl J Med. 2016;374:523–532. doi: 10.1056/NEJMoa1504327. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 37. Larsson SC, Traylor M, Malik R, Dichgans M, Burgess S, Markus HS. Modifiable pathways in Alzheimer's disease: Mendelian randomisation analysis. BMJ. 2017;359 doi: 10.1136/bmj.j5375. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 38. Kremen WS, Beck A, Elman JA, et al. Influence of young adult cognitive ability and additional education on later-life cognition. Proc Natl Acad Sci USA. 2019;116:2021–2026. doi: 10.1073/pnas.1811537116. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 39. Blacker D, Weuve J. Brain exercise and brain outcomes: does cognitive activity really work to maintain your brain? JAMA Psychiatry. 2018;75:703–704. doi: 10.1001/jamapsychiatry.2018.0656. [ DOI ] [ PubMed ] [ Google Scholar ]
- 40. Lee ATC, Richards M, Chan WC, Chiu HFK, Lee RSY, Lam LCW. Association of daily intellectual activities with lower risk of incident dementia among older Chinese adults. JAMA Psychiatry. 2018;75:697–703. doi: 10.1001/jamapsychiatry.2018.0657. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 41. Chan D, Shafto M, Kievit R, et al. Lifestyle activities in mid-life contribute to cognitive reserve in late-life, independent of education, occupation, and late-life activities. Neurobiol Aging. 2018;70:180–183. doi: 10.1016/j.neurobiolaging.2018.06.012. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 42. Staff RT, Hogan MJ, Williams DS, Whalley LJ. Intellectual engagement and cognitive ability in later life (the “use it or lose it” conjecture): longitudinal, prospective study. BMJ. 2018;363 doi: 10.1136/bmj.k4925. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 43. Kajitani S, Sakata K, McKenzie C. Occupation, retirement and cognitive functioning. Ageing Soc. 2017;37:1568–1596. [ Google Scholar ]
- 44. Xue B, Cadar D, Fleischmann M, et al. Effect of retirement on cognitive function: the Whitehall II cohort study. Eur J Epidemiol. 2018;33:989–1001. doi: 10.1007/s10654-017-0347-7. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 45. Meng A, Nexø MA, Borg V. The impact of retirement on age related cognitive decline - a systematic review. BMC Geriatr. 2017;17:160. doi: 10.1186/s12877-017-0556-7. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 46. Grotz C, Meillon C, Amieva H, et al. Why is later age at retirement beneficial for cognition? Results from a French population-based study. J Nutr Health Aging. 2016;20:514–519. doi: 10.1007/s12603-015-0599-4. [ DOI ] [ PubMed ] [ Google Scholar ]
- 47. Denier N, Clouston SAP, Richards M, Hofer SM. Retirement and cognition: a life course view. Adv Life Course Res. 2017;31:11–21. doi: 10.1016/j.alcr.2016.10.004. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 48. Clouston SA, Denier N. Mental retirement and health selection: analyses from the US health and retirement study. Soc Sci Med. 2017;178:78–86. doi: 10.1016/j.socscimed.2017.01.019. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 49. Rohwedder S, Willis RJ. Mental retirement. J Econ Perspect. 2010;24:119–138. doi: 10.1257/jep.24.1.119. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 50. Gates NJ, Sachdev PS, Fiatarone Singh MA, Valenzuela M. Cognitive and memory training in adults at risk of dementia: a systematic review. BMC Geriatr. 2011;11:55. doi: 10.1186/1471-2318-11-55. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 51. Gavelin HLA, Hallock H, Sabates J, Bahar-Fuchs A. Cognition-oriented treatments for older adults: a systematic overview of systematic reviews. Neuropsychol Rev. 2020;30:167–193. doi: 10.1007/s11065-020-09434-8. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 52. Kane RLBM, Fink HA, Brasure M, et al. Agency for Healthcare Research and Quality; Rockville, MD: 2017. Interventions to prevent age-related cognitive decline, mild cognitive impairment, and clinical Alzheimer's-type dementia. [ PubMed ] [ Google Scholar ]
- 53. Butler M, McCreedy E, Nelson VA, et al. Does cognitive training prevent cognitive decline? A systematic review. Ann Intern Med. 2018;168:63–68. doi: 10.7326/M17-1531. [ DOI ] [ PubMed ] [ Google Scholar ]
- 54. Gates NJ, Rutjes AW, Di Nisio M, et al. Computerised cognitive training for maintaining cognitive function in cognitively healthy people in midlife. Cochrane Database Syst Rev. 2019;3 doi: 10.1002/14651858.CD012278.pub2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 55. Hill NT, Mowszowski L, Naismith SL, Chadwick VL, Valenzuela M, Lampit A. Computerized cognitive training in older adults with mild cognitive impairment or dementia: a systematic review and meta-analysis. Am J Psychiatry. 2017;174:329–340. doi: 10.1176/appi.ajp.2016.16030360. [ DOI ] [ PubMed ] [ Google Scholar ]
- 56. Chandler MJ, Parks AC, Marsiske M, Rotblatt LJ, Smith GE. Everyday impact of cognitive interventions in mild cognitive impairment: a systematic review and meta-analysis. Neuropsychol Rev. 2016;26:225–251. doi: 10.1007/s11065-016-9330-4. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 57. Rovner BW, Casten RJ, Hegel MT, Leiby B. Preventing cognitive decline in Black individuals with mild cognitive impairment: a randomized clinical trial. JAMA Neurol. 2018;75:1487–1493. doi: 10.1001/jamaneurol.2018.2513. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 58. Loughrey DG, Kelly ME, Kelley GA, Brennan S, Lawlor BA. Association of age-related hearing loss with cognitive function, cognitive impairment, and dementia: a systematic review and meta-analysis. JAMA Otolaryngol Head Neck Surg. 2018;144:115–126. doi: 10.1001/jamaoto.2017.2513. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 59. Golub JS, Brickman AM, Ciarleglio AJ, Schupf N, Luchsinger JA. Association of subclinical hearing loss with cognitive performance. JAMA Otolaryngol Head Neck Surg. 2019;146:57–67. doi: 10.1001/jamaoto.2019.3375. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 60. Armstrong NM, An Y, Doshi J, et al. Association of midlife hearing impairment with late-life temporal lobe volume loss. JAMA Otolaryngol Head Neck Surg. 2019;145:794. doi: 10.1001/jamaoto.2019.1610. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 61. Amieva H, Ouvrard C, Meillon C, Rullier L, Dartigues JF. Death, depression, disability, and dementia associated with self-reported hearing problems: a 25-year study. J Gerontol A Biol Sci Med Sci. 2018;73:1383–1389. doi: 10.1093/gerona/glx250. [ DOI ] [ PubMed ] [ Google Scholar ]
- 62. Ray J, Popli G, Fell G. Association of cognition and age-related hearing impairment in the English longitudinal study of ageing. JAMA Otolaryngol Head Neck Surg. 2018;144:876–882. doi: 10.1001/jamaoto.2018.1656. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 63. Maharani A, Dawes P, Nazroo J, Tampubolon G, Pendleton N. Longitudinal relationship between hearing aid use and cognitive function in older Americans. J Am Geriatr Soc. 2018;66:1130–1136. doi: 10.1111/jgs.15363. [ DOI ] [ PubMed ] [ Google Scholar ]
- 64. Zanier ER, Bertani I, Sammali E, et al. Induction of a transmissible tau pathology by traumatic brain injury. Brain. 2018;141:2685–2699. doi: 10.1093/brain/awy193. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 65. Cao J, Gaamouch FE, Meabon JS, et al. ApoE4-associated phospholipid dysregulation contributes to development of tau hyper-phosphorylation after traumatic brain injury. Sci Rep. 2017;7 doi: 10.1038/s41598-017-11654-7. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 66. Bruns J, Jr, Hauser WA. The epidemiology of traumatic brain injury: a review. Epilepsia. 2003;44:2–10. doi: 10.1046/j.1528-1157.44.s10.3.x. [ DOI ] [ PubMed ] [ Google Scholar ]
- 67. Fann JR, Ribe AR, Pedersen HS, et al. Long-term risk of dementia among people with traumatic brain injury in Denmark: a population-based observational cohort study. Lancet Psychiatry. 2018;5:424–431. doi: 10.1016/S2215-0366(18)30065-8. [ DOI ] [ PubMed ] [ Google Scholar ]
- 68. Nordström A, Nordström P. Traumatic brain injury and the risk of dementia diagnosis: a nationwide cohort study. PLoS Med. 2018;15 doi: 10.1371/journal.pmed.1002496. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 69. Tolppanen AM, Taipale H, Hartikainen S. Head or brain injuries and Alzheimer's disease: a nested case-control register study. Alzheimers Dement. 2017;13:1371–1379. doi: 10.1016/j.jalz.2017.04.010. [ DOI ] [ PubMed ] [ Google Scholar ]
- 70. Barnes DE, Byers AL, Gardner RC, Seal KH, Boscardin WJ, Yaffe K. Association of mild traumatic brain injury with and without loss of consciousness with dementia in US military veterans. JAMA Neurol. 2018;75:1055–1061. doi: 10.1001/jamaneurol.2018.0815. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 71. Yaffe K, Lwi SJ, Hoang TD, et al. Military-related risk factors in female veterans and risk of dementia. Neurology. 2019;92:e205–e211. doi: 10.1212/WNL.0000000000006778. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 72. Redelmeier DA, Manzoor F, Thiruchelvam D. Association between statin use and risk of dementia after a concussion. JAMA Neurol. 2019;76:887. doi: 10.1001/jamaneurol.2019.1148. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 73. Smith DH, Johnson VE, Trojanowski JQ, Stewart W. Chronic traumatic encephalopathy - confusion and controversies. Nat Rev Neurol. 2019;15:179–183. doi: 10.1038/s41582-018-0114-8. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 74. Mackay DF, Russell ER, Stewart K, MacLean JA, Pell JP, Stewart W. Neurodegenerative disease mortality among former professional soccer players. N Engl J Med. 2019;381:1801–1808. doi: 10.1056/NEJMoa1908483. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 75. Pase MP, Beiser A, Enserro D, et al. Association of ideal cardiovascular health with vascular brain injury and incident dementia. Stroke. 2016;47:1201–1206. doi: 10.1161/STROKEAHA.115.012608. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 76. Lane CA, Barnes J, Nicholas JM, et al. Associations between blood pressure across adulthood and late-life brain structure and pathology in the neuroscience substudy of the 1946 British birth cohort (Insight 46): an epidemiological study. Lancet Neurol. 2019;18:942–952. doi: 10.1016/S1474-4422(19)30228-5. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 77. Walker KA, Sharrett AR, Wu A, et al. Association of midlife to late-life blood pressure patterns with incident dementia. JAMA. 2019;322:535–545. doi: 10.1001/jama.2019.10575. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 78. Williamson JD, Supiano MA, Applegate WB, et al. Intensive vs standard blood pressure control and cardiovascular disease outcomes in adults aged ≥75 years: a randomized clinical trial. JAMA. 2016;315:2673–2682. doi: 10.1001/jama.2016.7050. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 79. Williamson JD, Pajewski NM, Auchus AP, et al. Effect of intensive vs standard blood pressure control on probable dementia: a randomized clinical trial. JAMA. 2019;321:553–561. doi: 10.1001/jama.2018.21442. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 80. Yaffe K. Prevention of cognitive impairment with intensive systolic blood pressure control. JAMA. 2019;321:548–549. doi: 10.1001/jama.2019.0008. [ DOI ] [ PubMed ] [ Google Scholar ]
- 81. Peters R, Warwick J, Anstey KJ, Anderson CS. Blood pressure and dementia: what the SPRINT-MIND trial adds and what we still need to know. Neurology. 2019;92:1017–1018. doi: 10.1212/WNL.0000000000007543. [ DOI ] [ PubMed ] [ Google Scholar ]
- 82. Tully PJ, Hanon O, Cosh S, Tzourio C. Diuretic antihypertensive drugs and incident dementia risk: a systematic review, meta-analysis and meta-regression of prospective studies. J Hypertens. 2016;34:1027–1035. doi: 10.1097/HJH.0000000000000868. [ DOI ] [ PubMed ] [ Google Scholar ]
- 83. Ding J, Davis-Plourde KL, Sedaghat S, et al. Antihypertensive medications and risk for incident dementia and Alzheimer's disease: a meta-analysis of individual participant data from prospective cohort studies. Lancet Neurol. 2020;19:61–70. doi: 10.1016/S1474-4422(19)30393-X. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 84. Hussain S, Singh A, Rahman SO, Habib A, Najmi AK. Calcium channel blocker use reduces incident dementia risk in elderly hypertensive patients: a meta-analysis of prospective studies. Neurosci Lett. 2018;671:120–127. doi: 10.1016/j.neulet.2018.02.027. [ DOI ] [ PubMed ] [ Google Scholar ]
- 85. Peters R, Yasar S, Anderson CS, et al. An investigation of antihypertensive class, dementia, and cognitive decline: a meta-analysis. Neurology. 2019;94:e267–e281. doi: 10.1212/WNL.0000000000008732. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 86. McGuinness B, Craig D, Bullock R, Passmore P. Statins for the prevention of dementia. Cochrane Database Syst Rev. 2016;1 doi: 10.1002/14651858.CD003160.pub3. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 87. McNeil JJ, Woods RL, Nelson MR, et al. Effect of aspirin on disability-free survival in the healthy elderly. N Engl J Med. 2018;379:1499–1508. doi: 10.1056/NEJMoa1800722. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 88. Hersi M, Irvine B, Gupta P, Gomes J, Birkett N, Krewski D. Risk factors associated with the onset and progression of Alzheimer's disease: a systematic review of the evidence. Neurotoxicology. 2017;61:143–187. doi: 10.1016/j.neuro.2017.03.006. [ DOI ] [ PubMed ] [ Google Scholar ]
- 89. Zotcheva E, Bergh S, Selbæk G, et al. Midlife physical activity, psychological distress, and dementia risk: the HUNT study. J Alzheimers Dis. 2018;66:825–833. doi: 10.3233/JAD-180768. [ DOI ] [ PubMed ] [ Google Scholar ]
- 90. Hörder H, Johansson L, Guo X, et al. Midlife cardiovascular fitness and dementia: a 44-year longitudinal population study in women. Neurology. 2018;90:e1298–e1305. doi: 10.1212/WNL.0000000000005290. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 91. Kivimäki M, Singh-Manoux A, Pentti J, et al. Physical inactivity, cardiometabolic disease, and risk of dementia: an individual-participant meta-analysis. BMJ. 2019;365 doi: 10.1136/bmj.l1495. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 92. Saint-Maurice PF, Coughlan D, Kelly SP, et al. Association of leisure-time physical activity across the adult life course with all-cause and cause-specific mortality. JAMA Netw Open. 2019;2 doi: 10.1001/jamanetworkopen.2019.0355. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 93. Northey JM, Cherbuin N, Pumpa KL, Smee DJ, Rattray B. Exercise interventions for cognitive function in adults older than 50: a systematic review with meta-analysis. Br J Sports Med. 2018;52:154–160. doi: 10.1136/bjsports-2016-096587. [ DOI ] [ PubMed ] [ Google Scholar ]
- 94. Song D, Yu DSF, Li PWC, Lei Y. The effectiveness of physical exercise on cognitive and psychological outcomes in individuals with mild cognitive impairment: a systematic review and meta-analysis. Int J Nurs Stud. 2018;79:155–164. doi: 10.1016/j.ijnurstu.2018.01.002. [ DOI ] [ PubMed ] [ Google Scholar ]
- 95. de Souto Barreto P, Demougeot L, Vellas B, Rolland Y. Exercise training for preventing dementia, mild cognitive impairment, and clinically meaningful cognitive decline: a systematic review and meta-analysis. J Gerontol A Biol Sci Med Sci. 2018;73:1504–1511. doi: 10.1093/gerona/glx234. [ DOI ] [ PubMed ] [ Google Scholar ]
- 96. Barha CK, Davis JC, Falck RS, Nagamatsu LS, Liu-Ambrose T. Sex differences in exercise efficacy to improve cognition: a systematic review and meta-analysis of randomized controlled trials in older humans. Front Neuroendocrinol. 2017;46:71–85. doi: 10.1016/j.yfrne.2017.04.002. [ DOI ] [ PubMed ] [ Google Scholar ]
- 97. WHO . World Health Organization; Geneva: 2019. Risk reduction of cognitive decline and dementia: WHO guidelines. [ PubMed ] [ Google Scholar ]
- 98. Chatterjee S, Peters SA, Woodward M, et al. Type 2 diabetes as a risk factor for dementia in women compared with men: a pooled analysis of 2·3 million people comprising more than 100,000 cases of dementia. Diabetes Care. 2016;39:300–307. doi: 10.2337/dc15-1588. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 99. McMillan JM, Mele BS, Hogan DB, Leung AA. Impact of pharmacological treatment of diabetes mellitus on dementia risk: systematic review and meta-analysis. BMJ Open Diabetes Res Care. 2018;6 doi: 10.1136/bmjdrc-2018-000563. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 100. Sabia S, Fayosse A, Dumurgier J, et al. Association of ideal cardiovascular health at age 50 with incidence of dementia: 25 year follow-up of Whitehall II cohort study. BMJ. 2019;366 doi: 10.1136/bmj.l4414. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 101. Areosa Sastre A, Vernooij RW, González-Colaço Harmand M, Martínez G. Effect of the treatment of type 2 diabetes mellitus on the development of cognitive impairment and dementia. Cochrane Database Syst Rev. 2017;6 doi: 10.1002/14651858.CD003804.pub2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 102. Rehm J, Hasan OSM, Black SE, Shield KD, Schwarzinger M. Alcohol use and dementia: a systematic scoping review. Alzheimers Res Ther. 2019;11:1. doi: 10.1186/s13195-018-0453-0. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 103. Schwarzinger M, Pollock BG, Hasan OSM, et al. Contribution of alcohol use disorders to the burden of dementia in France 2008–13: a nationwide retrospective cohort study. Lancet Public Health. 2018;3:e124–e132. doi: 10.1016/S2468-2667(18)30022-7. [ DOI ] [ PubMed ] [ Google Scholar ]
- 104. Ilomaki J, Jokanovic N, Tan ECK, Lonnroos E. Alcohol consumption, dementia, and cognitive decline: an overview of systematic reviews. Curr Clin Pharmacol. 2015;10:204–212. doi: 10.2174/157488471003150820145539. [ DOI ] [ PubMed ] [ Google Scholar ]
- 105. Koch M, Fitzpatrick AL, Rapp SR, et al. Alcohol Consumption and risk of dementia and cognitive decline among older adults with or without mild cognitive impairment. JAMA Netw Open. 2019;2 doi: 10.1001/jamanetworkopen.2019.10319. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 106. Piumatti G, Moore SC, Berridge DM, Sarkar C, Gallacher J. The relationship between alcohol use and long-term cognitive decline in middle and late life: a longitudinal analysis using UK Biobank. J Public Health (Oxf) 2018;40:304–311. doi: 10.1093/pubmed/fdx186. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 107. Sabia S, Fayosse A, Dumurgier J, et al. Alcohol consumption and risk of dementia: 23 year follow-up of Whitehall II cohort study. BMJ. 2018;362 doi: 10.1136/bmj.k2927. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 108. Topiwala A, Allan CL, Valkanova V, et al. Moderate alcohol consumption as risk factor for adverse brain outcomes and cognitive decline: longitudinal cohort study. BMJ. 2017;357 doi: 10.1136/bmj.j2353. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 109. Albanese E, Launer LJ, Egger M, et al. Body mass index in midlife and dementia: systematic review and meta-regression analysis of 589,649 men and women followed in longitudinal studies. Alzheimers Dement (Amst) 2017;8:165–178. doi: 10.1016/j.dadm.2017.05.007. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 110. Veronese N, Facchini S, Stubbs B, et al. Weight loss is associated with improvements in cognitive function among overweight and obese people: a systematic review and meta-analysis. Neurosci Biobehav Rev. 2017;72:87–94. doi: 10.1016/j.neubiorev.2016.11.017. [ DOI ] [ PubMed ] [ Google Scholar ]
- 111. Chang CC, Zhao Y, Lee CW, Ganguli M. Smoking, death, and Alzheimer disease: a case of competing risks. Alzheimer Dis Assoc Disord. 2012;26:300–306. doi: 10.1097/WAD.0b013e3182420b6e. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 112. Debanne SM, Bielefeld RA, Cheruvu VK, Fritsch T, Rowland DY. Alzheimer's disease and smoking: bias in cohort studies. J Alzheimers Dis. 2007;11:313–321. doi: 10.3233/jad-2007-11308. [ DOI ] [ PubMed ] [ Google Scholar ]
- 113. Choi D, Choi S, Park SM. Effect of smoking cessation on the risk of dementia: a longitudinal study. Ann Clin Transl Neurol. 2018;5:1192–1199. doi: 10.1002/acn3.633. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 114. Oberg M, Jaakkola MS, Woodward A, Peruga A, Prüss-Ustün A. Worldwide burden of disease from exposure to second-hand smoke: a retrospective analysis of data from 192 countries. Lancet. 2011;377:139–146. doi: 10.1016/S0140-6736(10)61388-8. [ DOI ] [ PubMed ] [ Google Scholar ]
- 115. Pan X, Luo Y, Roberts AR. Secondhand Smoke and Women's Cognitive Function in China. Am J Epidemiol. 2018;187:911–918. doi: 10.1093/aje/kwx377. [ DOI ] [ PubMed ] [ Google Scholar ]
- 116. Prince MJA, Albanese E, Guerchet M, Prina M. Alzheimer's Disease International; London: 2014. The World Alzheimer Report 2014. Dementia and risk reduction. An analysis of protective and modifiable factors. [ Google Scholar ]
- 117. Almeida OP, Hankey GJ, Yeap BB, Golledge J, Flicker L. Depression as a modifiable factor to decrease the risk of dementia. Transl Psychiatry. 2017;7 doi: 10.1038/tp.2017.90. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 118. Kelly ME, Duff H, Kelly S, et al. The impact of social activities, social networks, social support and social relationships on the cognitive functioning of healthy older adults: a systematic review. Syst Rev. 2017;6:259. doi: 10.1186/s13643-017-0632-2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 119. Bartels C, Wagner M, Wolfsgruber S, Ehrenreich H, Schneider A. Alzheimer's disease neuroimaging I. Impact of SSRI therapy on risk of conversion rrom mild cognitive impairment to Alzheimer's dementia in individuals with previous depression. Am J Psychiatry. 2018;175:232–241. doi: 10.1176/appi.ajp.2017.17040404. [ DOI ] [ PubMed ] [ Google Scholar ]
- 120. Sommerlad A, Ruegger J, Singh-Manoux A, Lewis G, Livingston G. Marriage and risk of dementia: systematic review and meta-analysis of observational studies. J Neurol Neurosurg Psychiatry. 2018;89:231–238. doi: 10.1136/jnnp-2017-316274. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 121. Evans IEM, Martyr A, Collins R, Brayne C, Clare L. Social isolation and cognitive function in later life: a systematic review and meta-analysis. J Alzheimers Dis. 2019;70:S119–S144. doi: 10.3233/JAD-180501. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 122. Penninkilampi R, Casey AN, Singh MF, Brodaty H. The association between social engagement, loneliness, and risk of dementia: a systematic review and meta-analysis. J Alzheimers Dis. 2018;66:1619–1633. doi: 10.3233/JAD-180439. [ DOI ] [ PubMed ] [ Google Scholar ]
- 123. Sommerlad A, Sabia S, Singh-Manoux A, Lewis G, Livingston G. Association of social contact with dementia and cognition: 28-year follow-up of the Whitehall II cohort study. PLoS Med. 2019;16 doi: 10.1371/journal.pmed.1002862. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 124. Saito T, Murata C, Saito M, Takeda T, Kondo K. Influence of social relationship domains and their combinations on incident dementia: a prospective cohort study. J Epidemiol Community Health. 2018;72:7–12. doi: 10.1136/jech-2017-209811. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 125. Power MC, Adar SD, Yanosky JD, Weuve J. Exposure to air pollution as a potential contributor to cognitive function, cognitive decline, brain imaging, and dementia: a systematic review of epidemiologic research. Neurotoxicology. 2016;56:235–253. doi: 10.1016/j.neuro.2016.06.004. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 126. Chen H, Kwong JC, Copes R, et al. Living near major roads and the incidence of dementia, Parkinson's disease, and multiple sclerosis: a population-based cohort study. Lancet. 2017;389:718–726. doi: 10.1016/S0140-6736(16)32399-6. [ DOI ] [ PubMed ] [ Google Scholar ]
- 127. Oudin A, Segersson D, Adolfsson R, Forsberg B. Association between air pollution from residential wood burning and dementia incidence in a longitudinal study in Northern Sweden. PLoS One. 2018;13 doi: 10.1371/journal.pone.0198283. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 128. Oudin A, Forsberg B, Adolfsson AN, et al. Traffic-related air pollution and dementia incidence in northern Sweden: a longitudinal study. Environ Health Perspect. 2016;124:306–312. doi: 10.1289/ehp.1408322. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 129. Carey IM, Anderson HR, Atkinson RW, et al. Are noise and air pollution related to the incidence of dementia? A cohort study in London, England. BMJ Open. 2018;8 doi: 10.1136/bmjopen-2018-022404. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 130. Bowe B, Xie Y, Yan Y, Al-Aly Z. Burden of cause-specific mortality associated with PM2.5 air pollution in the United States. JAMA Netw Open. 2019;2 doi: 10.1001/jamanetworkopen.2019.15834. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 131. Spira AP, Gamaldo AA, An Y, et al. Self-reported sleep and β-amyloid deposition in community-dwelling older adults. JAMA Neurol. 2013;70:1537–1543. doi: 10.1001/jamaneurol.2013.4258. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 132. Macedo AC, Balouch S, Tabet N. Is sleep disruption a risk factor for Alzheimer's disease? J Alzheimers Dis. 2017;58:993–1002. doi: 10.3233/JAD-161287. [ DOI ] [ PubMed ] [ Google Scholar ]
- 133. Musiek ES, Xiong DD, Holtzman DM. Sleep, circadian rhythms, and the pathogenesis of Alzheimer disease. Exp Mol Med. 2015;47:e148. doi: 10.1038/emm.2014.121. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 134. Yaffe K, Falvey CM, Hoang T. Connections between sleep and cognition in older adults. Lancet Neurol. 2014;13:1017–1028. doi: 10.1016/S1474-4422(14)70172-3. [ DOI ] [ PubMed ] [ Google Scholar ]
- 135. Sindi S, Kåreholt I, Johansson L, et al. Sleep disturbances and dementia risk: a multicenter study. Alzheimers Dement. 2018;14:1235–1242. doi: 10.1016/j.jalz.2018.05.012. [ DOI ] [ PubMed ] [ Google Scholar ]
- 136. Irwin MR, Vitiello MV. Implications of sleep disturbance and inflammation for Alzheimer's disease dementia. Lancet Neurol. 2019;18:296–306. doi: 10.1016/S1474-4422(18)30450-2. [ DOI ] [ PubMed ] [ Google Scholar ]
- 137. Shi L, Chen SJ, Ma MY, et al. Sleep disturbances increase the risk of dementia: a systematic review and meta-analysis. Sleep Med Rev. 2018;40:4–16. doi: 10.1016/j.smrv.2017.06.010. [ DOI ] [ PubMed ] [ Google Scholar ]
- 138. Bubu OM, Brannick M, Mortimer J, et al. Sleep, Cognitive impairment, and Alzheimer's disease: a systematic review and meta-analysis. Sleep. 2017;40 doi: 10.1093/sleep/zsw032. [ DOI ] [ PubMed ] [ Google Scholar ]
- 139. Ohara T, Honda T, Hata J, et al. Association between daily sleep duration and risk of dementia and mortality in a Japanese community. J Am Geriatr Soc. 2018;66:1911–1918. doi: 10.1111/jgs.15446. [ DOI ] [ PubMed ] [ Google Scholar ]
- 140. Li J, Ogrodnik M, Kolachalama VB, Lin H, Au R. Assessment of the mid-life demographic and lifestyle risk factors of dementia using data from the framingham heart study offspring cohort. J Alzheimers Dis. 2018;63:1119–1127. doi: 10.3233/JAD-170917. [ DOI ] [ PubMed ] [ Google Scholar ]
- 141. Lutsey PL, Misialek JR, Mosley TH, et al. Sleep characteristics and risk of dementia and Alzheimer's disease: the atherosclerosis risk in communities study. Alzheimers Dement. 2018;14:157–166. doi: 10.1016/j.jalz.2017.06.2269. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 142. Lu Y, Sugawara Y, Zhang S, Tomata Y, Tsuji I. Changes in sleep duration and the risk of incident dementia in the elderly Japanese: the Ohsaki Cohort 2006 Study. Sleep. 2018;41 doi: 10.1093/sleep/zsy143. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 143. Richardson K, Mattishent K, Loke YK, et al. History of benzodiazepine prescriptions and risk of dementia: possible bias due to prevalent users and covariate measurement timing in a nested case-control study. Am J Epidemiol. 2019;188:1228–1236. doi: 10.1093/aje/kwz073. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 144. Bronskill SE, Campitelli MA, Iaboni A, et al. Low-dose trazodone, benzodiazepines, and fall-related injuries in nursing homes: a matched-cohort study. J Am Geriatr Soc. 2018;66:1963–1971. doi: 10.1111/jgs.15519. [ DOI ] [ PubMed ] [ Google Scholar ]
- 145. Pistollato F, Iglesias RC, Ruiz R, et al. Nutritional patterns associated with the maintenance of neurocognitive functions and the risk of dementia and Alzheimer's disease: a focus on human studies. Pharmacol Res. 2018;131:32–43. doi: 10.1016/j.phrs.2018.03.012. [ DOI ] [ PubMed ] [ Google Scholar ]
- 146. Morris MC, Wang Y, Barnes LL, Bennett DA, Dawson-Hughes B, Booth SL. Nutrients and bioactives in green leafy vegetables and cognitive decline: Prospective study. Neurology. 2018;90:e214–e222. doi: 10.1212/WNL.0000000000004815. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 147. Akbaraly TN, Singh-Manoux A, Dugravot A, Brunner EJ, Kivimäki M, Sabia S. Association of midlife diet with subsequent risk for dementia. JAMA. 2019;321:957–968. doi: 10.1001/jama.2019.1432. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 148. D'Cunha NM, Georgousopoulou EN, Dadigamuwage L, et al. Effect of long-term nutraceutical and dietary supplement use on cognition in the elderly: a 10-year systematic review of randomised controlled trials. Br J Nutr. 2018;119:280–298. doi: 10.1017/S0007114517003452. [ DOI ] [ PubMed ] [ Google Scholar ]
- 149. Rutjes AW, Denton DA, Di Nisio M, et al. Vitamin and mineral supplementation for maintaining cognitive function in cognitively healthy people in mid and late life. Cochrane Database Syst Rev. 2018;12 doi: 10.1002/14651858.CD011906.pub2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 150. McCleery J, Abraham RP, Denton DA, et al. Vitamin and mineral supplementation for preventing dementia or delaying cognitive decline in people with mild cognitive impairment. Cochrane Database Syst Rev. 2018;11 doi: 10.1002/14651858.CD011905.pub2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 151. Farina N, Isaac MGEKN, Clark AR, Rusted J, Tabet N. Vitamin E for Alzheimer's dementia and mild cognitive impairment. Cochrane Database Syst Rev. 2017;4 doi: 10.1002/14651858.CD002854.pub5. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 152. Soininen H, Solomon A, Visser PJ, et al. 24-month intervention with a specific multinutrient in people with prodromal Alzheimer's disease (LipiDiDiet): a randomised, double-blind, controlled trial. Lancet Neurol. 2017;16:965–975. doi: 10.1016/S1474-4422(17)30332-0. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 153. Loughrey DG, Lavecchia S, Brennan S, Lawlor BA, Kelly ME. The impact of the mediterranean diet on the cognitive functioning of healthy older adults: a systematic review and meta-analysis. Adv Nutr. 2017;8:571–586. doi: 10.3945/an.117.015495. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 154. Radd-Vagenas S, Duffy SL, Naismith SL, Brew BJ, Flood VM, Fiatarone Singh MA. Effect of the Mediterranean diet on cognition and brain morphology and function: a systematic review of randomized controlled trials. Am J Clin Nutr. 2018;107:389–404. doi: 10.1093/ajcn/nqx070. [ DOI ] [ PubMed ] [ Google Scholar ]
- 155. Rosenberg A, Ngandu T, Rusanen M, et al. Multidomain lifestyle intervention benefits a large elderly population at risk for cognitive decline and dementia regardless of baseline characteristics: the FINGER trial. Alzheimers Dement. 2018;14:263–270. doi: 10.1016/j.jalz.2017.09.006. [ DOI ] [ PubMed ] [ Google Scholar ]
- 156. Stephen R, Liu Y, Ngandu T, et al. Brain volumes and cortical thickness on MRI in the Finnish geriatric intervention study to prevent cognitive impairment and disability (FINGER) Alzheimers Res Ther. 2019;11:53. doi: 10.1186/s13195-019-0506-z. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 157. Richard E, Jongstra S, Soininen H, et al. Healthy ageing through internet counselling in the elderly: the HATICE randomised controlled trial for the prevention of cardiovascular disease and cognitive impairment. BMJ Open. 2016;6 doi: 10.1136/bmjopen-2015-010806. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 158. Richard E, Moll van Charante EP, Hoevenaar-Blom MP, et al. Healthy ageing through internet counselling in the elderly (HATICE): a multinational, randomised controlled trial. Lancet Digital Health. 2019;1:e424–e434. doi: 10.1016/S2589-7500(19)30153-0. [ DOI ] [ PubMed ] [ Google Scholar ]
- 159. NHS Digital Health Survey for England 2014: health, social care and lifestyles: summary of key findings, 2014. Dec 16, 2015. https://digital.nhs.uk/data-and-information/publications/statistical/health-survey-for-england/health-survey-for-england-2014
- 160. Xu W, Wang H, Wan Y, et al. Alcohol consumption and dementia risk: a dose-response meta-analysis of prospective studies. Eur J Epidemiol. 2017;32:31–42. doi: 10.1007/s10654-017-0225-3. [ DOI ] [ PubMed ] [ Google Scholar ]
- 161. Britton A, Ben-Shlomo Y, Benzeval M, Kuh D, Bell S. Life course trajectories of alcohol consumption in the United Kingdom using longitudinal data from nine cohort studies. BMC Med. 2015;13:47. doi: 10.1186/s12916-015-0273-z. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 162. Järvenpää T, Rinne JO, Koskenvuo M, Räihä I, Kaprio J. Binge drinking in midlife and dementia risk. Epidemiology. 2005;16:766–771. doi: 10.1097/01.ede.0000181307.30826.6c. [ DOI ] [ PubMed ] [ Google Scholar ]
- 163. Handing EP, Andel R, Kadlecova P, Gatz M, Pedersen NL. Midlife alcohol consumption and risk of dementia over 43 years of follow-up: a population-based study from the Swedish twin registry. J Gerontol A Biol Sci Med Sci. 2015;70:1248–1254. doi: 10.1093/gerona/glv038. [ DOI ] [ PubMed ] [ Google Scholar ]
- 164. Shor E, Roelfs D, Vang ZM. The “Hispanic mortality paradox” revisited: meta-analysis and meta-regression of life-course differentials in Latin American and Caribbean immigrants' mortality. Soc Sci Med. 2017;186:20–33. doi: 10.1016/j.socscimed.2017.05.049. [ DOI ] [ PubMed ] [ Google Scholar ]
- 165. Altman DG. Chapman and Hall/CRC press; London: 1990. Practical statistics for medical research. [ Google Scholar ]
- 166. Huang CH, Lin CW, Lee YC, et al. Is traumatic brain injury a risk factor for neurodegeneration? A meta-analysis of population-based studies. BMC Neurol. 2018;18:184. doi: 10.1186/s12883-018-1187-0. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 167. Zhang J, Yu KF. What's the relative risk? A method of correcting the odds ratio in cohort studies of common outcomes. JAMA. 1998;280:1690–1691. doi: 10.1001/jama.280.19.1690. [ DOI ] [ PubMed ] [ Google Scholar ]
- 168. Chu SF, Chiu WT, Lin HW, Chiang YH, Liou TH. Hazard ratio and repeat injury for dementia in patients with and without a history of traumatic brain injury: a population-based secondary data analysis in Taiwan. Asia Pac J Public Health. 2016;28:519–527. doi: 10.1177/1010539516662956. [ DOI ] [ PubMed ] [ Google Scholar ]
- 173. Frost RB, Farrer TJ, Primosch M, Hedges DW. Prevalence of traumatic brain injury in the general adult population: a meta-analysis. Neuroepidemiology. 2013;40:154–159. doi: 10.1159/000343275. [ DOI ] [ PubMed ] [ Google Scholar ]
- 174. Chen H, Kwong JC, Copes R, et al. Exposure to ambient air pollution and the incidence of dementia: a population-based cohort study. Environ Int. 2017;108:271–277. doi: 10.1016/j.envint.2017.08.020. [ DOI ] [ PubMed ] [ Google Scholar ]
- 175. Lourida I, Hannon E, Littlejohns TJ, et al. Association of lifestyle and genetic risk with incidence of dementia. JAMA. 2019;322:430. doi: 10.1001/jama.2019.9879. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 176. Solomon A, Turunen H, Ngandu T, et al. Effect of the apolipoprotein E genotype on cognitive change during a multidomain lifestyle intervention: a subgroup analysis of a randomized clinical trial. JAMA Neurol. 2018;75:462–470. doi: 10.1001/jamaneurol.2017.4365. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 177. Licher S, Ahmad S, Karamujić-Čomić H, et al. Genetic predisposition, modifiable-risk-factor profile and long-term dementia risk in the general population. Nat Med. 2019;25:1364–1369. doi: 10.1038/s41591-019-0547-7. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 178. Rockwood K, Wallace LMK, Davis DH. Genetic predisposition and modifiable risks for late-life dementia. Nat Med. 2019;25:1331–1332. doi: 10.1038/s41591-019-0575-3. [ DOI ] [ PubMed ] [ Google Scholar ]
- 179. Ritchie K, Carrière I, Ritchie CW, Berr C, Artero S, Ancelin ML. Designing prevention programmes to reduce incidence of dementia: prospective cohort study of modifiable risk factors. BMJ. 2010;341 doi: 10.1136/bmj.c3885. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 180. American Psychiatric Association . American Psychiatric Press; Washington, DC: 1996. DSM-IV sourcebook. [ Google Scholar ]
- 181. American Psychiatric Association . 5th edn. American Psychiatric Association; Washington, DC: 2013. DSM-5 Task Force. Diagnostic and statistical manual of mental disorders: DSM-5. [ Google Scholar ]
- 182. Roberts RO, Aakre JA, Kremers WK, et al. Prevalence and outcomes of amyloid positivity among persons without dementia in a longitudinal, population-based setting. JAMA Neurol. 2018;75:970–979. doi: 10.1001/jamaneurol.2018.0629. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 183. Rice L, Bisdas S. The diagnostic value of FDG and amyloid PET in Alzheimer's disease-a systematic review. Eur J Radiol. 2017;94:16–24. doi: 10.1016/j.ejrad.2017.07.014. [ DOI ] [ PubMed ] [ Google Scholar ]
- 184. Dang C, Harrington KD, Lim YY, et al. Relationship between amyloid-β positivity and progression to mild cognitive impairment or dementia over 8 years in cognitively normal older adults. J Alzheimers Dis. 2018;65:1313–1325. doi: 10.3233/JAD-180507. [ DOI ] [ PubMed ] [ Google Scholar ]
- 185. Brookmeyer R, Abdalla N. Estimation of lifetime risks of Alzheimer's disease dementia using biomarkers for preclinical disease. Alzheimers Dement. 2018;14:981–988. doi: 10.1016/j.jalz.2018.03.005. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 186. Jack CR, Jr, Wiste HJ, Therneau TM, et al. Associations of amyloid, tau, and neurodegeneration biomarker profiles with rates of memory decline among individuals without dementia. JAMA. 2019;321:2316–2325. doi: 10.1001/jama.2019.7437. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 187. Rabinovici GD, Gatsonis C, Apgar C, et al. Association of amyloid positron emission tomography with subsequent change in clinical management among Medicare beneficiaries with mild cognitive impairment or dementia. JAMA. 2019;321:1286–1294. doi: 10.1001/jama.2019.2000. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 188. Nakamura A, Kaneko N, Villemagne VL, et al. High performance plasma amyloid-β biomarkers for Alzheimer's disease. Nature. 2018;554:249–254. doi: 10.1038/nature25456. [ DOI ] [ PubMed ] [ Google Scholar ]
- 189. Ovod V, Ramsey KN, Mawuenyega KG, et al. Amyloid β concentrations and stable isotope labeling kinetics of human plasma specific to central nervous system amyloidosis. Alzheimers Dement. 2017;13:841–849. doi: 10.1016/j.jalz.2017.06.2266. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 190. Janelidze S, Stomrud E, Palmqvist S, et al. Plasma β-amyloid in Alzheimer's disease and vascular disease. Sci Rep. 2016;6 doi: 10.1038/srep26801. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 191. Blennow K, Mattsson N, Schöll M, Hansson O, Zetterberg H. Amyloid biomarkers in Alzheimer's disease. Trends Pharmacol Sci. 2015;36:297–309. doi: 10.1016/j.tips.2015.03.002. [ DOI ] [ PubMed ] [ Google Scholar ]
- 192. Herukka SK, Simonsen AH, Andreasen N, et al. Recommendations for cerebrospinal fluid Alzheimer's disease biomarkers in the diagnostic evaluation of mild cognitive impairment. Alzheimers Dement. 2017;13:285–295. doi: 10.1016/j.jalz.2016.09.009. [ DOI ] [ PubMed ] [ Google Scholar ]
- 193. Byrne LM, Rodrigues FB, Blennow K, et al. Neurofilament light protein in blood as a potential biomarker of neurodegeneration in Huntington's disease: a retrospective cohort analysis. Lancet Neurol. 2017;16:601–609. doi: 10.1016/S1474-4422(17)30124-2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 194. Disanto G, Barro C, Benkert P, et al. Serum neurofilament light: a biomarker of neuronal damage in multiple sclerosis. Ann Neurol. 2017;81:857–870. doi: 10.1002/ana.24954. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 195. Mattsson N, Andreasson U, Zetterberg H, Blennow K. Association of plasma neurofilament light with neurodegeneration in patients with Alzheimer disease. JAMA Neurol. 2017;74:557–566. doi: 10.1001/jamaneurol.2016.6117. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 196. Patnode CD, Perdue LA, Rossom RC, et al. The Agency for Healthcare Research and Quality Publication; Rockville, MD: 2020. Screening for cognitive impairment in older adults: an evidence update for the US Preventive Services Task Force [Internet] [ PubMed ] [ Google Scholar ]
- 197. Fowler NR, Perkins AJ, Gao S, Sachs GA, Boustani MA. Risks and benefits of screening for dementia in primary care: the Indiana University cognitive health outcomes investigation of the comparative effectiveness of dementia screening (IU CHOICE) trial. J Am Geriatr Soc. 2019;68:535–543. doi: 10.1111/jgs.16247. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 198. Fowler NR, Perkins AJ, Gao S, Sachs GA, Uebelhor AK, Boustani MA. Patient characteristics associated with screening positive for Alzheimer's disease and related dementia. Clin Interv Aging. 2018;13:1779–1785. doi: 10.2147/CIA.S164957. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 199. NICE . National Institute for Health and Care Excellence; London: 2018. Dementia: Assessment, management and support for people living with dementia and their carers. [ PubMed ] [ Google Scholar ]
- 200. Birks JS, Harvey RJ. Donepezil for dementia due to Alzheimer's disease. Cochrane Database Syst Rev. 2018;6 doi: 10.1002/14651858.CD001190.pub3. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 201. Walsh S, King E, Brayne C. France removes state funding for dementia drugs. BMJ. 2019;367 doi: 10.1136/bmj.l6930. [ DOI ] [ PubMed ] [ Google Scholar ]
- 202. Egan MF, Kost J, Tariot PN, et al. Randomized trial of verubecestat for mild-to-moderate Alzheimer's disease. N Engl J Med. 2018;378:1691–1703. doi: 10.1056/NEJMoa1706441. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 203. Lawlor B, Segurado R, Kennelly S, et al. Nilvadipine in mild to moderate Alzheimer disease: a randomised controlled trial. PLoS Med. 2018;15 doi: 10.1371/journal.pmed.1002660. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 204. Andrews M, Tousi B, Sabbagh MN. 5HT6 antagonists in the treatment of Alzheimer's dementia: current progress. Neurol Ther. 2018;7:51–58. doi: 10.1007/s40120-018-0095-y. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 205. Voytyuk I, De Strooper B, Chávez-Gutiérrez L. Modulation of γ- and β-secretases as early prevention against Alzheimer's disease. Biol Psychiatry. 2018;83:320–327. doi: 10.1016/j.biopsych.2017.08.001. [ DOI ] [ PubMed ] [ Google Scholar ]
- 206. Egan MF, Kost J, Voss T, et al. Randomized trial of verubecestat for prodromal Alzheimer's disease. N Engl J Med. 2019;380:1408–1420. doi: 10.1056/NEJMoa1812840. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 207. Bahar-Fuchs A, Martyr A, Goh AM, Sabates J, Clare L. Cognitive training for people with mild to moderate dementia. Cochrane Database Syst Rev. 2019;3 doi: 10.1002/14651858.CD013069.pub2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 208. Lamb SE, Sheehan B, Atherton N, et al. Dementia and physical activity (DAPA) trial of moderate to high intensity exercise training for people with dementia: randomised controlled trial. BMJ. 2018;361 doi: 10.1136/bmj.k1675. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 209. Teri L, Logsdon RG, McCurry AM, Pike KC, McGough EL. Translating an evidence-based multicomponent intervention for older adults with dementia and caregivers. Gerontologist. 2020;60:548–557. doi: 10.1093/geront/gny122. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 210. Ehrenberg AJ, Suemoto CK, França Resende EP, et al. Neuropathologic correlates of psychiatric symptoms in Alzheimer's disease. J Alzheimers Dis. 2018;66:115–126. doi: 10.3233/JAD-180688. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 211. Donegan K, Fox N, Black N, Livingston G, Banerjee S, Burns A. Trends in diagnosis and treatment for people with dementia in the UK from 2005 to 2015: a longitudinal retrospective cohort study. Lancet Public Health. 2017;2:e149–e156. doi: 10.1016/S2468-2667(17)30031-2. [ DOI ] [ PubMed ] [ Google Scholar ]
- 212. Thyrian JR, Hertel J, Wucherer D, et al. Effectiveness and safety of dementia care management in primary care: a randomized clinical trial. JAMA Psychiatry. 2017;74:996–1004. doi: 10.1001/jamapsychiatry.2017.2124. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 213. Gitlin LN, Arthur P, Piersol C, et al. Targeting behavioral symptoms and functional decline in dementia: a randomized clinical trial. J Am Geriatr Soc. 2018;66:339–345. doi: 10.1111/jgs.15194. [ DOI ] [ PubMed ] [ Google Scholar ]
- 214. Dudas R, Malouf R, McCleery J, Dening T. Antidepressants for treating depression in dementia. Cochrane Database Syst Rev. 2018;8 doi: 10.1002/14651858.CD003944.pub2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 215. Orgeta V, Tabet N, Nilforooshan R, Howard R. Efficacy of antidepressants for depression in Alzheimer's disease: systematic review and meta-analysis. J Alzheimers Dis. 2017;58:725–733. doi: 10.3233/JAD-161247. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 216. Panca M, Livingston G, Barber J, et al. Healthcare resource utilisation and costs of agitation in people with dementia living in care homes in England - the managing agitation and raising quality of life in dementia (MARQUE) study. PLoS One. 2019;14 doi: 10.1371/journal.pone.0211953. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 217. Livingston G, Barber J, Marston L, et al. Prevalence of and associations with agitation in residents with dementia living in care homes: MARQUE cross-sectional study. BJPsych Open. 2017;3:171–178. doi: 10.1192/bjpo.bp.117.005181. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 218. Cooper C, Marston L, Barber J, et al. Do care homes deliver person-centred care? A cross-sectional survey of staff-reported abusive and positive behaviours towards residents from the MARQUE (managing agitation and raising quality of life) English national care home survey. PLoS One. 2018;13 doi: 10.1371/journal.pone.0193399. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 219. Ballard C, Corbett A, Orrell M, et al. Impact of person-centred care training and person-centred activities on quality of life, agitation, and antipsychotic use in people with dementia living in nursing homes: A cluster-randomised controlled trial. PLoS Med. 2018;15 doi: 10.1371/journal.pmed.1002500. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 220. Lichtwarck B, Selbaek G, Kirkevold Ø, et al. Targeted interdisciplinary model for evaluation and treatment of neuropsychiatric symptoms: a cluster randomized controlled trial. Am J Geriatr Psychiatry. 2018;26:25–38. doi: 10.1016/j.jagp.2017.05.015. [ DOI ] [ PubMed ] [ Google Scholar ]
- 221. Schneider LS, Dagerman K, Insel PS. Efficacy and adverse effects of atypical antipsychotics for dementia: meta-analysis of randomized, placebo-controlled trials. Am J Geriatr Psychiatry. 2006;14:191–210. doi: 10.1097/01.JGP.0000200589.01396.6d. [ DOI ] [ PubMed ] [ Google Scholar ]
- 222. Kales HC, Gitlin LN, Lyketsos CG. Management of neuropsychiatric symptoms of dementia in clinical settings: recommendations from a multidisciplinary expert panel. J Am Geriatr Soc. 2014;62:762–769. doi: 10.1111/jgs.12730. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 223. Livingston G, Barber J, Marston L, et al. Clinical and cost-effectiveness of the managing agitation and raising quality of life (MARQUE) intervention for agitation in people with dementia in care homes: a single-blind, cluster-randomised controlled trial. Lancet Psychiatry. 2019;6:293–304. doi: 10.1016/S2215-0366(19)30045-8. [ DOI ] [ PubMed ] [ Google Scholar ]
- 224. Kales HC, Lyketsos CG, Miller EM, Ballard C. Management of behavioral and psychological symptoms in people with Alzheimer's disease: an international Delphi consensus. Int Psychogeriatr. 2018;31:83–90. doi: 10.1017/S1041610218000534. [ DOI ] [ PubMed ] [ Google Scholar ]
- 225. Van Leeuwen E, Petrovic M, van Driel ML, et al. Withdrawal versus continuation of long-term antipsychotic drug use for behavioural and psychological symptoms in older people with dementia. Cochrane Database Syst Rev. 2018;3 doi: 10.1002/14651858.CD007726.pub3. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 226. Cossette B, Bruneau MA, Couturier Y, et al. Optimizing practices, use, care and services-antipsychotics (OPUS-AP) in long-term care centers in Quebec, Canada: a strategy for best practices. J Am Med Dir Assoc. 2019 doi: 10.1016/j.jamda.2019.08.027. [ DOI ] [ PubMed ] [ Google Scholar ]
- 227. Cohen-Mansfield J. Do reports on personal preferences of persons with dementia predict their responses to group activities? Dement Geriatr Cogn Disord. 2018;46:100–108. doi: 10.1159/000491746. [ DOI ] [ PubMed ] [ Google Scholar ]
- 228. Cohen-Mansfield J. The impact of group activities and their content on persons with dementia attending them. Alzheimers Res Ther. 2018;10:37. doi: 10.1186/s13195-018-0357-z. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 229. Ruthirakuhan MT, Herrmann N, Abraham EH, Chan S, Lanctôt KL. Pharmacological interventions for apathy in Alzheimer's disease. Cochrane Database Syst Rev. 2018;5 doi: 10.1002/14651858.CD012197.pub2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 230. Kinnunen KM, Vikhanova A, Livingston G. The management of sleep disorders in dementia: an update. Curr Opin Psychiatry. 2017;30:491–497. doi: 10.1097/YCO.0000000000000370. [ DOI ] [ PubMed ] [ Google Scholar ]
- 231. Livingston G, Barber JA, Kinnunen KM, et al. DREAMS-START (Dementia RElAted Manual for Sleep; STrAtegies for RelaTives) for people with dementia and sleep disturbances: a single-blind feasibility and acceptability randomized controlled trial. Int Psychogeriatr. 2018;31:251–265. doi: 10.1017/S1041610218000753. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 232. Maust DT, Kales HC, McCammon RJ, Blow FC, Leggett A, Langa KM. Distress associated with dementia-related psychosis and agitation in relation to healthcare utilization and costs. Am J Geriatr Psychiatry. 2017;25:1074–1082. doi: 10.1016/j.jagp.2017.02.025. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 233. Livingston G, Manela M, O'Keeffe A, et al. Clinical effectiveness of the START (STrAtegies for RelaTives) psychological intervention for family carers and the effects on the cost of care for people with dementia: 6-year follow-up of a randomised controlled trial. Br J Psychiatry. 2020;216:35–42. doi: 10.1192/bjp.2019.160. [ DOI ] [ PubMed ] [ Google Scholar ]
- 234. Guterman EL, Allen IE, Josephson SA, et al. Association between caregiver depression and emergency department use among patients with dementia. JAMA Neurol. 2019;76:1166–1173. doi: 10.1001/jamaneurol.2019.1820. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 235. Clare L, Kudlicka A, Oyebode JR, et al. Goal-oriented cognitive rehabilitation for early-stage Alzheimer's and related dementias: the GREAT RCT. Health Technol Assess. 2019;23:1–242. doi: 10.3310/hta23100. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 236. Scott I, Cooper C, Leverton M, et al. Effects of nonpharmacological interventions on functioning of people living with dementia at home: a systematic review of randomised controlled trials. Int J Geriatr Psychiatry. 2019;34:1386–1402. doi: 10.1002/gps.5127. [ DOI ] [ PubMed ] [ Google Scholar ]
- 237. Sharma S, Mueller C, Stewart R, et al. Predictors of falls and fractures leading to hospitalization in people with dementia: a representative cohort study. J Am Med Dir Assoc. 2018;19:607–612. doi: 10.1016/j.jamda.2018.03.009. [ DOI ] [ PubMed ] [ Google Scholar ]
- 238. Cooper C, Lodwick R, Walters K, et al. Inequalities in receipt of mental and physical healthcare in people with dementia in the UK. Age Ageing. 2017;46:393–400. doi: 10.1093/ageing/afw208. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 239. Fereshtehnejad SM, Garcia-Ptacek S, Religa D, et al. Dental care utilization in patients with different types of dementia: a longitudinal nationwide study of 58,037 individuals. Alzheimers Dement. 2018;14:10–19. doi: 10.1016/j.jalz.2017.05.004. [ DOI ] [ PubMed ] [ Google Scholar ]
- 240. Bunn F, Goodman C, Reece Jones P, et al. What works for whom in the management of diabetes in people living with dementia: a realist review. BMC Med. 2017;15:141. doi: 10.1186/s12916-017-0909-2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 241. Banerjee S, Farina N, Daley S, et al. How do we enhance undergraduate healthcare education in dementia? A review of the role of innovative approaches and development of the Time for Dementia Programme. Int J Geriatr Psychiatry. 2017;32:68–75. doi: 10.1002/gps.4602. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 242. Browne J, Edwards DA, Rhodes KM, Brimicombe DJ, Payne RA. Association of comorbidity and health service usage among patients with dementia in the UK: a population-based study. BMJ Open. 2017;7 doi: 10.1136/bmjopen-2016-012546. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 243. Poblador-Plou B, Calderón-Larrañaga A, Marta-Moreno J, et al. Comorbidity of dementia: a cross-sectional study of primary care older patients. BMC Psychiatry. 2014;14:84. doi: 10.1186/1471-244X-14-84. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 244. Rait G, Walters K, Bottomley C, Petersen I, Iliffe S, Nazareth I. Survival of people with clinical diagnosis of dementia in primary care: cohort study. BMJ. 2010;341 doi: 10.1136/bmj.c3584. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 245. Bauer K, Schwarzkopf L, Graessel E, Holle R. A claims data-based comparison of comorbidity in individuals with and without dementia. BMC Geriatr. 2014;14:10. doi: 10.1186/1471-2318-14-10. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 246. Bunn F, Burn A-M, Goodman C, et al. Comorbidity and dementia: a scoping review of the literature. BMC Med. 2014;12:192. doi: 10.1186/s12916-014-0192-4. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 247. Bennett HQ, Norton S, Bunn F, et al. The impact of dementia on service use by individuals with a comorbid health condition: a comparison of two cross-sectional analyses conducted approximately 10 years apart. BMC Med. 2018;16:114. doi: 10.1186/s12916-018-1105-8. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 248. Melis RJ, Marengoni A, Rizzuto D, et al. The influence of multimorbidity on clinical progression of dementia in a population-based cohort. PLoS One. 2013;8 doi: 10.1371/journal.pone.0084014. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 249. Martín-García S, Rodríguez-Blázquez C, Martínez-López I, Martínez-Martín P, Forjaz MJ. Comorbidity, health status, and quality of life in institutionalized older people with and without dementia. Int Psychogeriatr. 2013;25:1077–1084. doi: 10.1017/S1041610213000458. [ DOI ] [ PubMed ] [ Google Scholar ]
- 250. Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 251. Verity R, Okell LC, Dorigatti I, et al. Estimates of the severity of coronavirus disease 2019: a model-based analysis. Lancet Infect Dis. 2020;20:669–677. doi: 10.1016/S1473-3099(20)30243-7. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 252. Guan WJ, Liang WH, Zhao Y, et al. Comorbidity and its impact on 1590 patients with Covid-19 in China: a nationwide analysis. Eur Respir J. 2020;55 doi: 10.1183/13993003.00547-2020. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 253. Office for National Statistics Impact of coronavirus in care homes in England: 26 May to 19 June 2020. 2020. https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases/articles/impactofcoronavirusincarehomesinenglandvivaldi/26mayto19june2020
- 254. Livingston G, Weidner W. COVID-19 and dementia: difficult decisions about hospital admission and triage. 2020. https://www.alz.co.uk/news/adi-releases-position-paper-on-covid-19-and-dementia
- 255. Troyer EA, Kohn JN, Hong S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain Behav Immun. 2020;87:34–39. doi: 10.1016/j.bbi.2020.04.027. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 256. Ritchie K, Chan D, Watermeyer T. The neurological consequences of the COVID-19 epidemic: collateral damage? Brain Communications. 2020 doi: 10.1093/braincomms/fcaa069. published online May 28. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 257. Nicol GE, Piccirillo JF, Mulsant BH, Lenze EJ. Action at a distance: geriatric research during a pandemic. J Am Geriatr Soc. 2020;68:922–925. doi: 10.1111/jgs.16443. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 258. Wang H, Li T, Barbarino P, et al. Dementia care during COVID-19. Lancet. 2020;395:1190–1191. doi: 10.1016/S0140-6736(20)30755-8. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 259. Lau-Ng R, Caruso LB, Perls TT. COVID-19 deaths in long term care facilities - a critical piece of the pandemic puzzle. J Am Geriatr Soc. 2020 doi: 10.1111/jgs.16669. published online June 5. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 260. Comas-Herrera A, Zalakain J, Litwin C, Hsu AT, Fefnandez-Plotka JL. Mortality associated with COVID-19 outbreaks in care homes: early international evidence. 2020. https://ltccovid.org/2020/04/12/mortality-associated-with-covid-19-outbreaks-in-care-homes-early-international-evidence/
- 261. McMichael TM, Currie DW, Clark S, et al. Epidemiology of Covid-19 in a long-term care facility in King County, Washington. N Engl J Med. 2020;382:2005–2011. doi: 10.1056/NEJMoa2005412. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 262. CarePort Health COVID-19 report. 2020. https://careporthealth.com/wp-content/uploads/2020/05/COVID-19-Report-3.pdf
- 263. Zhan M, Qin Y, Xue X, Zhu S. Death from Covid-19 of 23 Health care workers in China. N Engl J Med. 2020;382:2267–2268. doi: 10.1056/NEJMc2005696. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 264. Chen T, Wu D, Chen H, et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. 2020;368 doi: 10.1136/bmj.m1091. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 265. White N, Leurent B, Lord K, Scott S, Jones L, Sampson EL. The management of behavioural and psychological symptoms of dementia in the acute general medical hospital: a longitudinal cohort study. Int J Geriatr Psychiatry. 2017;32:297–305. doi: 10.1002/gps.4463. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 266. Malone DC, McLaughlin TP, Wahl PM, et al. Burden of Alzheimer's disease and association with negative health outcomes. Am J Manag Care. 2009;15:481–488. [ PubMed ] [ Google Scholar ]
- 267. Sager MA, Rudberg MA, Jalaluddin M, et al. Hospital admission risk profile (HARP): identifying older patients at risk for functional decline following acute medical illness and hospitalization. J Am Geriatr Soc. 1996;44:251–257. doi: 10.1111/j.1532-5415.1996.tb00910.x. [ DOI ] [ PubMed ] [ Google Scholar ]
- 268. Phelan EA, Borson S, Grothaus L, Balch S, Larson EB. Association of incident dementia with hospitalizations. JAMA. 2012;307:165–172. doi: 10.1001/jama.2011.1964. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 269. Zhao Y, Kuo TC, Weir S, Kramer MS, Ash AS. Healthcare costs and utilization for Medicare beneficiaries with Alzheimer's. BMC Health Serv Res. 2008;8:108. doi: 10.1186/1472-6963-8-108. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 270. Bynum JP, Rabins PV, Weller W, Niefeld M, Anderson GF, Wu AW. The relationship between a dementia diagnosis, chronic illness, Medicare expenditures, and hospital use. J Am Geriatr Soc. 2004;52:187–194. doi: 10.1111/j.1532-5415.2004.52054.x. [ DOI ] [ PubMed ] [ Google Scholar ]
- 271. Shepherd H, Livingston G, Chan J, Sommerlad A. Hospitalisation rates and predictors in people with dementia: a systematic review and meta-analysis. BMC Med. 2019;17:130. doi: 10.1186/s12916-019-1369-7. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 272. Zhu CW, Cosentino S, Ornstein K, Gu Y, Andrews H, Stern Y. Use and cost of hospitalization in dementia: longitudinal results from a community-based study. Int J Geriatr Psychiatry. 2015;30:833–841. doi: 10.1002/gps.4222. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 273. Zhu CW, Cosentino S, Ornstein KA, Gu Y, Andrews H, Stern Y. interactive effects of dementia severity and comorbidities on medicare expenditures. J Alzheimers Dis. 2017;57:305–315. doi: 10.3233/JAD-161077. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 274. MacNeil Vroomen J, Bosmans JE, Eekhout I, et al. The cost-effectiveness of two forms of case management compared to a control group for persons with dementia and their informal caregivers from a societal perspective. PLoS One. 2016;11 doi: 10.1371/journal.pone.0160908. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 275. Intrator O, Zinn J, Mor V. Nursing home characteristics and potentially preventable hospitalizations of long-stay residents. J Am Geriatr Soc. 2004;52:1730–1736. doi: 10.1111/j.1532-5415.2004.52469.x. [ DOI ] [ PubMed ] [ Google Scholar ]
- 276. Phelan EA, Debnam KJ, Anderson LA, Owens SB. A systematic review of intervention studies to prevent hospitalizations of community-dwelling older adults with dementia. Med Care. 2015;53:207–213. doi: 10.1097/MLR.0000000000000294. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 277. Naylor MD, Brooten D, Campbell R, et al. Comprehensive discharge planning and home follow-up of hospitalized elders: a randomized clinical trial. JAMA. 1999;281:613–620. doi: 10.1001/jama.281.7.613. [ DOI ] [ PubMed ] [ Google Scholar ]
- 278. Handley M, Bunn F, Goodman C. Dementia-friendly interventions to improve the care of people living with dementia admitted to hospitals: a realist review. BMJ Open. 2017;7 doi: 10.1136/bmjopen-2016-015257. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 279. Department of Health . Department of Health; Leeds: 2012. Using the Commissioning for Quality and Innovation (CQUIN) payment framework Guidance on new national goals for 2012–13. [ Google Scholar ]
- 280. Wilkinson T, Ly A, Schnier C, et al. Identifying dementia cases with routinely collected health data: a systematic review. Alzheimers Dement. 2018;14:1038–1051. doi: 10.1016/j.jalz.2018.02.016. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 281. Sommerlad A, Perera G, Singh-Manoux A, Lewis G, Stewart R, Livingston G. Accuracy of general hospital dementia diagnoses in England: sensitivity, specificity, and predictors of diagnostic accuracy 2008-2013. Alzheimers Dement. 2018;14:933–943. doi: 10.1016/j.jalz.2018.02.012. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 282. Ryan DJ, O'Regan NA, Caoimh RÓ, et al. Delirium in an adult acute hospital population: predictors, prevalence and detection. BMJ Open. 2013;3 doi: 10.1136/bmjopen-2012-001772. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 283. Jackson TA, MacLullich AM, Gladman JR, Lord JM, Sheehan B. Undiagnosed long-term cognitive impairment in acutely hospitalised older medical patients with delirium: a prospective cohort study. Age Ageing. 2016;45:493–499. doi: 10.1093/ageing/afw064. [ DOI ] [ PubMed ] [ Google Scholar ]
- 284. Davis DHJ, Muniz-Terrera G, Keage HAD, et al. Association of delirium with cognitive decline in late life: a neuropathologic study of 3 population-based cohort studies. JAMA Psychiatry. 2017;74:244–251. doi: 10.1001/jamapsychiatry.2016.3423. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 285. James BD, Wilson RS, Capuano AW, et al. Cognitive decline after elective and nonelective hospitalizations in older adults. Neurology. 2019;92:e690–e699. doi: 10.1212/WNL.0000000000006918. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 286. Fong TG, Davis D, Growdon ME, Albuquerque A, Inouye SK. The interface between delirium and dementia in elderly adults. Lancet Neurol. 2015;14:823–832. doi: 10.1016/S1474-4422(15)00101-5. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 287. Siddiqi N, Harrison JK, Clegg A, et al. Interventions for preventing delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2016;3 doi: 10.1002/14651858.CD005563.pub3. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 288. Neufeld KJ, Yue J, Robinson TN, Inouye SK, Needham DM. Antipsychotic medication for prevention and treatment of delirium in hospitalized adults: a systematic review and meta-analysis. J Am Geriatr Soc. 2016;64:705–714. doi: 10.1111/jgs.14076. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 289. Barbateskovic M, Krauss SR, Collet MO, et al. Pharmacological interventions for prevention and management of delirium in intensive care patients: a systematic overview of reviews and meta-analyses. BMJ Open. 2019;9 doi: 10.1136/bmjopen-2018-024562. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 290. Hshieh TT, Yang T, Gartaganis SL, Yue J, Inouye SK. Hospital elder life program: systematic review and meta-analysis of effectiveness. Am J Geriatr Psychiatry. 2018;26:1015–1033. doi: 10.1016/j.jagp.2018.06.007. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 291. Wu YC, Tseng PT, Tu YK, et al. Association of delirium response and safety of pharmacological interventions for the management and prevention of delirium: a network meta-analysis. JAMA Psychiatry. 2019;76:526–535. doi: 10.1001/jamapsychiatry.2018.4365. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 292. Goldberg SE, Bradshaw LE, Kearney FC, et al. Care in specialist medical and mental health unit compared with standard care for older people with cognitive impairment admitted to general hospital: randomised controlled trial (NIHR TEAM trial) BMJ. 2013;347 doi: 10.1136/bmj.f4132. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 293. Kolanowski A, Fick D, Litaker M, et al. Effect of cognitively stimulating activities on symptom management of delirium superimposed on dementia: A randomized controlled trial. J Am Geriatr Soc. 2016;64:2424–2432. doi: 10.1111/jgs.14511. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 294. Burry L, Mehta S, Perreault MM, et al. Antipsychotics for treatment of delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. 2018;6 doi: 10.1002/14651858.CD005594.pub3. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 295. Yu A, Wu S, Zhang Z, et al. Cholinesterase inhibitors for the treatment of delirium in non-ICU settings. Cochrane Database Syst Rev. 2018;6 doi: 10.1002/14651858.CD012494.pub2. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 296. Lonergan E, Luxenberg J, Areosa Sastre A, Wyller T. Benzodiazepines for delirium. Cochrane Database Syst Rev. 2009;4 doi: 10.1002/14651858.CD006379.pub2. [ DOI ] [ PubMed ] [ Google Scholar ]
- 297. The American Geriatrics Society 2015 Beers Criteria Update Expert Panel American Geriatrics Society 2015 Updated Beers Criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2015;63:2227–2246. doi: 10.1111/jgs.13702. [ DOI ] [ PubMed ] [ Google Scholar ]
- 298. Jackson TA, Gladman JR, Harwood RH, et al. Challenges and opportunities in understanding dementia and delirium in the acute hospital. PLoS Med. 2017;14 doi: 10.1371/journal.pmed.1002247. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 299. Boyle PA, Yu L, Leurgans SE, et al. Attributable risk of Alzheimer's dementia attributed to age-related neuropathologies. Ann Neurol. 2019;85:114–124. doi: 10.1002/ana.25380. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 300. Boyle PA, Yu L, Wilson RS, Leurgans SE, Schneider JA, Bennett DA. Person-specific contribution of neuropathologies to cognitive loss in old age. Ann Neurol. 2018;83:74–83. doi: 10.1002/ana.25123. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 301. Wallace L, Theou O, Rockwood K, Andrew MK. Relationship between frailty and Alzheimer's disease biomarkers: a scoping review. Alzheimers Dement (Amst) 2018;10:394–401. doi: 10.1016/j.dadm.2018.05.002. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 302. Aworinde J, Werbeloff N, Lewis G, Livingston G, Sommerlad A. Dementia severity at death: a register-based cohort study. BMC Psychiatry. 2018;18:355. doi: 10.1186/s12888-018-1930-5. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 303. Meeussen K, Van den Block L, Echteld M, et al. Older people dying with dementia: a nationwide study. Int Psychogeriatr. 2012;24:1581–1591. doi: 10.1017/S1041610212000865. [ DOI ] [ PubMed ] [ Google Scholar ]
- 304. Sampson EL, Candy B, Davis S, et al. Living and dying with advanced dementia: A prospective cohort study of symptoms, service use and care at the end of life. Palliat Med. 2018;32:668–681. doi: 10.1177/0269216317726443. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 305. Rosemond C, Hanson LC, Zimmerman S. Goals of care or goals of trust? how family members perceive goals for dying nursing home residents. J Palliat Med. 2017;20:360–365. doi: 10.1089/jpm.2016.0271. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 306. Hanson LC, Zimmerman S, Song MK, et al. Effect of the Goals of care intervention for advanced dementia: a randomized clinical trial. JAMA Intern Med. 2017;177:24–31. doi: 10.1001/jamainternmed.2016.7031. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 307. Dening KH, King M, Jones L, Sampson EL. Healthcare decision-making: past present and future, in light of a diagnosis of dementia. Int J Palliat Nurs. 2017;23:4–11. doi: 10.12968/ijpn.2017.23.1.4. [ DOI ] [ PubMed ] [ Google Scholar ]
- 308. Gaster B, Larson EB, Curtis JR. Advance directives for dementia: meeting a unique challenge. JAMA. 2017;318:2175–2176. doi: 10.1001/jama.2017.16473. [ DOI ] [ PubMed ] [ Google Scholar ]
- 309. Harrison Dening K, King M, Jones L, Vickestaff V, Sampson EL. Advance care planning in dementia: do family carers know the treatment preferences of people with early dementia? PLoS One. 2016;11 doi: 10.1371/journal.pone.0159056. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 310. Brazil K, Carter G, Cardwell C, et al. Effectiveness of advance care planning with family carers in dementia nursing homes: a paired cluster randomized controlled trial. Palliat Med. 2018;32:603–612. doi: 10.1177/0269216317722413. [ DOI ] [ PubMed ] [ Google Scholar ]
- 311. Shah SM, Carey IM, Harris T, DeWilde S, Victor CR, Cook DG. The mental health and mortality impact of death of a partner with dementia. Int J Geriatr Psychiatry. 2016;31:929–937. doi: 10.1002/gps.4411. [ DOI ] [ PubMed ] [ Google Scholar ]
- 312. Moore KJ, Davis S, Gola A, et al. Experiences of end of life amongst family carers of people with advanced dementia: longitudinal cohort study with mixed methods. BMC Geriatr. 2017;17:135. doi: 10.1186/s12877-017-0523-3. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 313. Agar M, Luckett T, Luscombe G, et al. Effects of facilitated family case conferencing for advanced dementia: a cluster randomised clinical trial. PLoS One. 2017;12 doi: 10.1371/journal.pone.0181020. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 314. Luckett T, Chenoweth L, Phillips J, et al. A facilitated approach to family case conferencing for people with advanced dementia living in nursing homes: perceptions of palliative care planning coordinators and other health professionals in the IDEAL study. Int Psychogeriatr. 2017;29:1713–1722. doi: 10.1017/S1041610217000977. [ DOI ] [ PubMed ] [ Google Scholar ]
- 315. Pickett J, Bird C, Ballard C, et al. A roadmap to advance dementia research in prevention, diagnosis, intervention, and care by 2025. Int J Geriatr Psychiatry. 2018;33:900–906. doi: 10.1002/gps.4868. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
Uncited References
- 169. Abner EL, Nelson PT, Schmitt FA, et al. Self-reported head injury and risk of late-life impairment and AD pathology in an AD center cohort. Dement Geriatr Cogn Dis. 2014;37:294–306. doi: 10.1159/000355478. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 170. Gardner RC, Burke JF, Nettiksimmons J, Kaup A, Barnes DE, Yaffe K. Dementia risk after traumatic brain injury vs nonbrain trauma: the role of age and severity. JAMA Neurology. 2014;71:1490–1497. doi: 10.1001/jamaneurol.2014.2668. [ DOI ] [ PMC free article ] [ PubMed ] [ Google Scholar ]
- 171. Nordström P, Michaëlsson K, Gustafson Y, Nordström A. Traumatic brain injury and young onset dementia: a nationwide cohort study. Ann Neurol. 2014;75:374–381. doi: 10.1002/ana.24101. [ DOI ] [ PubMed ] [ Google Scholar ]
- 172. Wang H, Lin S, Sung P, et al. Population based study on patients with traumatic brain injury suggests increased risk of dementia. J Neurol Neurosurg Psychiatry. 2012;83:1080–1085. doi: 10.1136/jnnp-2012-302633. [ DOI ] [ PubMed ] [ Google Scholar ]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
- View on publisher site
- PDF (1.8 MB)
- Collections
Similar articles
Cited by other articles, links to ncbi databases.
- Download .nbib .nbib
- Format: AMA APA MLA NLM
Add to Collections
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- My Bibliography
- Collections
- Citation manager
Save citation to file
Email citation, add to collections.
- Create a new collection
- Add to an existing collection
Add to My Bibliography
Your saved search, create a file for external citation management software, your rss feed.
- Search in PubMed
- Search in NLM Catalog
- Add to Search
Diagnosis and Management of Dementia: Review
Affiliations.
- 1 Rush Alzheimer's Disease Center, Rush University Medical Center, Chicago, Illinois.
- 2 Department of Neurological Sciences, Rush University Medical Center, Chicago, Illinois.
- 3 Department of Family Medicine, Rush University Medical Center, Chicago, Illinois.
- PMID: 31638686
- PMCID: PMC7462122
- DOI: 10.1001/jama.2019.4782
Importance: Worldwide, 47 million people live with dementia and, by 2050, the number is expected to increase to 131 million.
Observations: Dementia is an acquired loss of cognition in multiple cognitive domains sufficiently severe to affect social or occupational function. In the United States, Alzheimer disease, one cause of dementia, affects 5.8 million people. Dementia is commonly associated with more than 1 neuropathology, usually Alzheimer disease with cerebrovascular pathology. Diagnosing dementia requires a history evaluating for cognitive decline and impairment in daily activities, with corroboration from a close friend or family member, in addition to a thorough mental status examination by a clinician to delineate impairments in memory, language, attention, visuospatial cognition such as spatial orientation, executive function, and mood. Brief cognitive impairment screening questionnaires can assist in initiating and organizing the cognitive assessment. However, if the assessment is inconclusive (eg, symptoms present, but normal examination findings), neuropsychological testing can help determine whether dementia is present. Physical examination may help identify the etiology of dementia. For example, focal neurologic abnormalities suggest stroke. Brain neuroimaging may demonstrate structural changes including, but not limited to, focal atrophy, infarcts, and tumor, that may not be identified on physical examination. Additional evaluation with cerebrospinal fluid assays or genetic testing may be considered in atypical dementia cases, such as age of onset younger than 65 years, rapid symptom onset, and/or impairment in multiple cognitive domains but not episodic memory. For treatment, patients may benefit from nonpharmacologic approaches, including cognitively engaging activities such as reading, physical exercise such as walking, and socialization such as family gatherings. Pharmacologic approaches can provide modest symptomatic relief. For Alzheimer disease, this includes an acetylcholinesterase inhibitor such as donepezil for mild to severe dementia, and memantine (used alone or as an add-on therapy) for moderate to severe dementia. Rivastigmine can be used to treat symptomatic Parkinson disease dementia.
Conclusions and relevance: Alzheimer disease currently affects 5.8 million persons in the United States and is a common cause of dementia, which is usually accompanied by other neuropathology, often cerebrovascular disease such as brain infarcts. Causes of dementia can be diagnosed by medical history, cognitive and physical examination, laboratory testing, and brain imaging. Management should include both nonpharmacologic and pharmacologic approaches, although efficacy of available treatments remains limited.
PubMed Disclaimer
Similar articles
- Treatment of Alzheimer disease. Winslow BT, Onysko MK, Stob CM, Hazlewood KA. Winslow BT, et al. Am Fam Physician. 2011 Jun 15;83(12):1403-12. Am Fam Physician. 2011. PMID: 21671540
- Screening for Cognitive Impairment in Older Adults: An Evidence Update for the U.S. Preventive Services Task Force [Internet]. Patnode CD, Perdue LA, Rossom RC, Rushkin MC, Redmond N, Thomas RG, Lin JS. Patnode CD, et al. Rockville (MD): Agency for Healthcare Research and Quality (US); 2020 Feb. Report No.: 19-05257-EF-1. Rockville (MD): Agency for Healthcare Research and Quality (US); 2020 Feb. Report No.: 19-05257-EF-1. PMID: 32129963 Free Books & Documents. Review.
- Screening for Cognitive Impairment in Older Adults: An Evidence Update for the U.S. Preventive Services Task Force [Internet]. Lin JS, O'Connor E, Rossom RC, Perdue LA, Burda BU, Thompson M, Eckstrom E. Lin JS, et al. Rockville (MD): Agency for Healthcare Research and Quality (US); 2013 Nov. Report No.: 14-05198-EF-1. Rockville (MD): Agency for Healthcare Research and Quality (US); 2013 Nov. Report No.: 14-05198-EF-1. PMID: 24354019 Free Books & Documents. Review.
- Dementia -- Caring, Ethics, Ethnical and Economical Aspects: A Systematic Review [Internet]. Swedish Council on Health Technology Assessment. Swedish Council on Health Technology Assessment. Stockholm: Swedish Council on Health Technology Assessment (SBU); 2008 Jun. SBU Assessment No. 172. Stockholm: Swedish Council on Health Technology Assessment (SBU); 2008 Jun. SBU Assessment No. 172. PMID: 28876770 Free Books & Documents. Review.
- Withdrawal or continuation of cholinesterase inhibitors or memantine or both, in people with dementia. Parsons C, Lim WY, Loy C, McGuinness B, Passmore P, Ward SA, Hughes C. Parsons C, et al. Cochrane Database Syst Rev. 2021 Feb 3;2(2):CD009081. doi: 10.1002/14651858.CD009081.pub2. Cochrane Database Syst Rev. 2021. PMID: 35608903 Free PMC article. Review.
- Global burden of dementia in younger people: an analysis of data from the 2021 Global Burden of Disease Study. Li Z, Yang Y, Liu Y, Wang X, Ping F, Xu L, Zhang H, Li W, Li Y. Li Z, et al. EClinicalMedicine. 2024 Oct 3;77:102868. doi: 10.1016/j.eclinm.2024.102868. eCollection 2024 Nov. EClinicalMedicine. 2024. PMID: 39416391 Free PMC article.
- Investigating Causal Associations between the Gut Microbiota and Dementia: A Mendelian Randomization Study. Xiong ZY, Li HM, Qiu CS, Tang XL, Liao DQ, Du LY, Lai SM, Huang HX, Zhang BY, Kuang L, Li ZH. Xiong ZY, et al. Nutrients. 2024 Sep 30;16(19):3312. doi: 10.3390/nu16193312. Nutrients. 2024. PMID: 39408279 Free PMC article.
- Effects of traditional Chinese medicine on outcomes and costs of dementia care: results from a retrospective real-world study. Weng YX, Yang CC, Hsu WC, Kuo RN. Weng YX, et al. Aging Clin Exp Res. 2024 Oct 12;36(1):204. doi: 10.1007/s40520-024-02858-9. Aging Clin Exp Res. 2024. PMID: 39395084 Free PMC article.
- Hearing loss in patients with dementia in Saudi Arabia. Alqahtani L, Alotaibi L, Alkhunein J, Alduaiji R, Alqadiri R, Alibrahim F, binSalih S, Balubaid H. Alqahtani L, et al. Neurosciences (Riyadh). 2024 Oct;29(4):262-269. doi: 10.17712/nsj.2024.4.20240030. Neurosciences (Riyadh). 2024. PMID: 39379090 Free PMC article.
- Mortality rate of pulmonary infection in senile dementia patients: A systematic review and meta-analysis. Yao J, Liu S, Chen Q. Yao J, et al. Medicine (Baltimore). 2024 Sep 20;103(38):e39816. doi: 10.1097/MD.0000000000039816. Medicine (Baltimore). 2024. PMID: 39312341 Free PMC article.
- Alzheimer’s Disease International. World Alzheimer Report 2015: the Global Impact of Dementia. An Analyses of Prevalence, incidence, Cost and Trends. https://www.alz.co.uk/research/WorldAlzheimerReport2015.pdf . Accessed March 20, 2018.
- Wimo A, Jönsson L, Bond J, Prince M, Winblad B; Alzheimer Disease International. The worldwide economic impact of dementia 2010. Alzheimers Dement. 2013. January;9(1):1–11. e3. doi: 10.1016/j.jalz.2012.11.006. - DOI - PubMed
- Goodman RA, Lochner KA, Thambisetty M, Wingo TS, Posner SF, Ling SM. Prevalence of dementia subtypes in United States Medicare fee-for-service beneficiaries, 2011–2013. Alzheimers Dement. 2017. January;13(1):28–37. doi: 10.1016/j.jalz.2016.04.002. Epub 2016 May 10. - DOI - PMC - PubMed
- Hebert LE, Weuve J, Scherr PA, Evans DA. Alzheimer disease in the United States (2010–2050) estimated using the 2010 census. Neurology. 2013. May 7;80(19):1778–1783. doi: 10.1212/WNL.0b013e31828726f5. Epub 2013 Feb 6. - DOI - PMC - PubMed
- U.S. Department of Health and Human services. Centers for Disease Control and Prevention. National Center for Health Statistics. Health, United States, 2015: With Special Feature on Racial and Ethnic Health Disparities. Hyattsville, MD: 2016. https://www.cdc.gov/nchs/data/hus/hus15.pdf . Accessed March 20, 2018. - PubMed
Publication types
- Search in MeSH
Related information
- Cited in Books
- PubChem Compound (MeSH Keyword)
Grants and funding
- P30 AG010161/AG/NIA NIH HHS/United States
- R01 AG040039/AG/NIA NIH HHS/United States
- R01 NS084965/NS/NINDS NIH HHS/United States
- RF1 AG059621/AG/NIA NIH HHS/United States
LinkOut - more resources
Full text sources.
- Europe PubMed Central
- Ovid Technologies, Inc.
- PubMed Central
- Silverchair Information Systems
Other Literature Sources
- The Lens - Patent Citations
- MedlinePlus Health Information
- Citation Manager
NCBI Literature Resources
MeSH PMC Bookshelf Disclaimer
The PubMed wordmark and PubMed logo are registered trademarks of the U.S. Department of Health and Human Services (HHS). Unauthorized use of these marks is strictly prohibited.

An official website of the United States government
Here's how you know
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
NIH releases 2022 dementia research progress report
Alzheimer's Disease
NIH has released Advancing Alzheimer’s Disease and Related Dementias Research for All Populations: Prevent. Diagnose. Treat. Care. (PDF, 17M), a 2022 scientific progress report.
The report features science advances and related efforts made between March 2021 and early 2022 in areas including drug development, lifestyle interventions, biomarker research, and more. It provides a comprehensive overview of the meaningful progress researchers are making to address the enormous health care challenges of Alzheimer’s and related dementia diseases.
This year’s progress report was preceded by the Fiscal Year 2024 Professional Judgment Budget for Alzheimer’s Disease and Related Dementias, announced in late July. Looking Forward: Opportunities to Accelerate Alzheimer’s and Related Dementias Research (PDF, 9M) provides an estimate of the funds needed to enable NIH to fully pursue scientific opportunities to inform effective prevention, treatment, and care of those living with these diseases.
Over the past year, NIH has conducted and funded remarkable Alzheimer’s and related dementias research that is bringing us closer to effective prevention, diagnostics, and treatments and improved care for the people living with these conditions, along with better support for care partners. With continued federal support and collaboration among researchers, clinicians, people living with dementia, and their care partners and families, the future holds hope and promise.
nia.nih.gov
An official website of the National Institutes of Health
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Review Article
- Published: 18 May 2023
Social participation and risk of developing dementia
- Andrew Sommerlad ORCID: orcid.org/0000-0002-8895-7055 1 , 2 ,
- Mika Kivimäki ORCID: orcid.org/0000-0002-4699-5627 1 , 3 ,
- Eric B. Larson 4 ,
- Susanne Röhr 5 , 6 , 7 ,
- Kokoro Shirai 8 ,
- Archana Singh-Manoux ORCID: orcid.org/0000-0002-1244-5037 1 , 9 &
- Gill Livingston ORCID: orcid.org/0000-0001-6741-5516 1 , 2
Nature Aging volume 3 , pages 532–545 ( 2023 ) Cite this article
15k Accesses
36 Citations
374 Altmetric
Metrics details
The increasing number of people with dementia globally illustrates the urgent need to reduce dementia’s scale and impact. Lifetime social participation may affect dementia risk by increasing cognitive reserve, and through brain maintenance by reducing stress and improving cerebrovascular health. It may therefore have important implications for individual behavior and public health policy aimed at reducing dementia burden. Observational study evidence indicates that greater social participation in midlife and late life is associated with 30–50% lower subsequent dementia risk, although some of this may not be causal. Social participation interventions have led to improved cognition but, partly due to short follow-up and small numbers of participants, no reduction in risk of dementia. We summarize the evidence linking social participation with dementia, discuss potential mechanisms by which social participation is likely to reduce and mitigate the impact of neuropathology in the brain, and consider the implications for future clinical and policy dementia prevention interventions.
Similar content being viewed by others

Social engagement and cognitive function among middle-aged and older adults: gender-specific findings from the Korean longitudinal study of aging (2008–2018)
A meta-analysis of loneliness and risk of dementia using longitudinal data from >600,000 individuals
Social determinants and lifestyle factors for brain health: implications for risk reduction of cognitive decline and dementia
Dementia is highly prevalent with 50 million people globally estimated to currently have dementia, and is forecast to grow markedly so that by 2050 there will be 152 million people living with dementia worldwide 1 . Dementia has a substantial impact on individuals and their families and is reported to be the most feared condition in older people 2 . Health and social care costs are estimated to be US $263 billion per year globally, with this figure forecast to rise to US $1.6 trillion by 2050, accounting for 11–17% of all health care spending 3 . The public health impact of dementia calls attention to the need to identify approaches to delay or prevent dementia. Projections indicate that delaying dementia onset by 5 years would halve prevalence 4 , and a 3-year delay would reduce dementia cost by one-third 5 .
There is suggestive evidence that changes in behaviors and improvements in health during the life course affect the risk of developing dementia. This is supported by studies of successive generations of older people showing that age-standardized dementia incidence and prevalence rates in several high-income countries have changed 6 , attributed to better health and education 7 . For example, in the UK Cognitive Function and Ageing Studies, a 20% reduced incidence rate was observed between 1990 and 2010 (ref. 8 ) and in the US Health and Retirement Study, the prevalence rate decreased by 13% between 2000 and 2012 (ref. 7 ). These findings have been replicated in other settings, although the opposite trend has been found in Japan 9 . Dementia rates may have declined through adoption of a healthier lifestyle, such as reduced tobacco smoking 10 or alcohol intake, and earlier and more effective treatment of hypertension 11 and diabetes 12 , or better access to education 7 . Conversely, an increasingly Western diet and physical inactivity leading to metabolic syndrome may account for Japan’s increased dementia prevalence rate 13 .
The overall evidence was summarized in the 2020 Lancet Commission for Dementia Prevention, Intervention and Care, which estimated that 12 potentially modifiable risk factors—lower education, hypertension, hearing impairment, smoking, obesity, depression, physical inactivity, diabetes, alcohol intake, poor sleep, air pollution and low social participation—may account for 40% of worldwide dementia risk 14 . The proportion may be higher in low-income and middle-income countries where these risk factors overall are more common, although there is variation between and within countries in the prevalence of individual risk factors 15 , 16 .
Social participation has attracted interest as a potential factor influencing general health and well-being, as well as specifically cognitive health and risk of dementia; it may promote healthy lifestyles, reduce stress and build cognitive health. Furthermore, longstanding societal changes and those related to the coronavirus disease 2019 (COVID-19) pandemic have increased the focus on social isolation and loneliness 17 , 18 . The 2019 World Health Organization’s (WHO) guidelines on risk reduction for cognitive decline and dementia 19 did not include social participation, but subsequent research findings in this area necessitate reexamination of the role of social participation.
Understanding the association between different aspects of social participation with dementia risk is important as it could guide measures to improve social participation behaviors at the individual and societal levels. While societal changes may contribute to social isolation and reducing social participation, there is little research evidence on social participation trends over past decades. Some studies show that formal social participation increased in older adults in the USA 20 , and loneliness levels in Swedish older adults were stable between 1992 and 2014 (ref. 21 ). The COVID-19 pandemic brought focus on social isolation and participation as measures to curb disease spread led to social distancing in most countries whereby reducing social participation was mandatory, with subsequent effects on mental health 22 . Social participation has not returned to pre-pandemic levels, and the effect of these changes on the health of older adults and on the risk of dementia remains unclear. However, some evidence suggests that people with dementia deteriorated more rapidly during the pandemic 23 , emphasizing the importance of considering whether and how to target increasing social participation.
Social participation is defined as ‘a person’s involvement in activities providing interactions with others in community life and in important shared spaces … based on the societal context and what individuals want and is meaningful to them’ 24 . We will use the term here to encompass engagement of individuals in sociable leisure activities (with focus on activities that are undertaken with other people), contact with social networks and their satisfaction with this participation. Definitions of this and related constructs are in Table 1 .
In this Review, we aim to elucidate the links between social participation and dementia risk to inform current and future approaches to prevent dementia through clinical interventions and policy measures to increase social participation. We do so by examining the current observational and interventional evidence on social participation and the associated risk of dementia and the mechanisms underlying these links. We conclude this Review by summarizing the implications for future treatments and policy.
Association between social participation and dementia
There is growing epidemiological evidence for an association between aspects of social participation and risk of dementia. These observational studies have examined social participation in midlife or late life and followed participants to compare dementia incidence, usually ascertained from clinical examination using diagnostic criteria or health records, between those with more and less social participation. Diagnosis of the cause of dementia (for example, Alzheimer’s disease or vascular dementia) is typically not ascertained with high-quality examination in these studies. Methods used to assess social participation have largely relied on self-report questionnaires completed by participants. These include validated scales such as the Berkman–Syme Index 25 for aspects of social network contact, and the UCLA scale 26 for assessing loneliness. Several unvalidated scales, created for the purposes of a study, have been used to assess participation in social activities.
Social activity engagement
Accumulating evidence suggests that having less engagement in social activities is associated with higher relative risk of dementia 27 , 28 , 29 , 30 (Fig. 1 ). Such studies generally combine activities into a composite activity scale regardless of how social these activities are; some involve other people (board games or group discussions), while others are solitary (crosswords and reading).
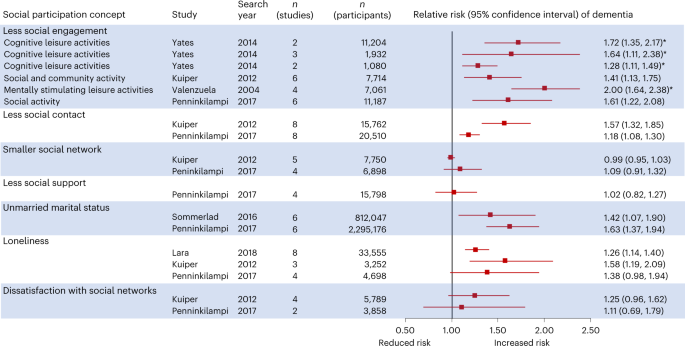
Summary of the evidence from six systematic reviews 27 , 37 , 38 , 39 , 44 , 144 about the association in epidemiological studies of different aspects of social participation with dementia risk. Relative risks indicate the risk of dementia associated with having less social participation; those marked with an asterisk were inverted from the published paper for consistency.
However, a major challenge of observational research of risk factors for dementia is the potential for reverse causation underpinning observed associations, so follow-up duration must be considered in this literature. Pathological changes develop decades before the diagnostic threshold for dementia is reached, and some symptoms of dementia precede diagnosis by many years during this preclinical phase 31 , 32 . Thus, establishing that a particular characteristic or behavior is present before dementia diagnosis does not confirm its status as a risk factor, as that characteristic may instead be an early consequence of dementia pathology arising during dementia development.
Many studies have a short follow-up; for example, five of the seven studies in one review of the effect of cognitive leisure activities had less than 6 years of an interval between activity measurement and dementia assessment, and all these studies found significant associations, whereas the remaining two studies, with 9 and 12 years of follow-up did not 27 . To reduce the risk of reverse causation, studies require a long interval between measurement of social participation and assessment for dementia and should exclude people with impaired cognitive status at baseline. Recent studies in the UK, Japan and China with 8–15 years of follow-up have linked more social engagement, participation in cognitive, intellectual and social activities, membership of social organizations and community engagement with lower dementia risk 28 , 29 , 30 , 33 , 34 .
Further insight can be gained from examination of trajectories of engagement in social activities leading up to dementia diagnosis using data from studies with repeated assessments of social participation. In two such studies of UK adults, not participating in social or leisure activities was associated with higher risk of dementia only with less than 10 years of follow-up 35 , 36 . And two studies found that declining social activity engagement was associated with higher dementia risk 28 , 35 suggesting that this decline occurs during the preclinical phase of dementia.
Altogether, less frequent engagement with social and leisure activities is linked with elevated subsequent dementia risk, but this association is at least partly due to reverse causation. Social activity engagement frequently co-occurs with cognitively stimulating activity, and social contact with others, so the specific contribution of activity engagement is unclear.
Social networks and social contact
Less frequent social contact with others is shown in two reviews to be associated with higher risk of dementia 37 , 38 (Fig. 1 .). Being unmarried, taken as a marker for having less lifetime social contact with others, is associated with elevated risk of dementia in two systematic reviews 38 , 39 (Fig. 1 ). One study found that having children was associated with lower dementia risk 40 , partly linked to marital status. Marital and parental status may also influence individuals’ ability to manage their health, and access informal care, which may account for some of this association. By contrast, simply having a smaller social network has not been found to be associated with dementia risk 37 , 38 , suggesting that interacting with people frequently, rather than interacting with more people, is important.
Duration of follow-up may again explain the inconsistency in results from these studies; seven of the eight studies in one of the reviews had less than 4 years of follow-up, and so it is impossible to judge the direction of causation 37 . However, a subsequent study with a longer duration of follow-up found that less frequent social contact in UK adults at age 60, particularly with friends rather than relatives, was associated with greater dementia risk and worse cognitive decline 15 years later 41 . Two studies of participants in the UK Biobank study with 9 years 42 and 12 years 43 of mean follow-up found that dementia risk was higher for those who were more socially isolated, with this finding consistent across different levels of genetic risk 42 and linked to lower gray matter volume 43 .
Hence, there is strong evidence that frequent social contact with others, but not having a larger social network, reduces the risk of dementia.
Subjective aspects of social participation
Three reviews found that reporting loneliness is associated with greater dementia risk compared to people who were not lonely 37 , 38 , 44 (Fig. 1 ). Five subsequent studies of loneliness in the USA over 10 years 45 , 46 , 47 , over 14 years in Sweden 48 and over 5 years in Japan 49 have found it to be associated with 34–91% elevated dementia risk. Some of these studies found the association to persist after adjusting for potential confounders including social isolation 46 , 49 , 50 . However, studies of UK adults found that loneliness was not independently associated with dementia risk and that the unadjusted association was probably mediated by depression symptoms 42 , 43 . Reporting less social support was also unrelated to dementia risk in a systematic review 38 , although a recent study of Korean adults with 10 years of follow-up found low emotional, but not practical, support was associated with elevated risk of all-cause dementia and Alzheimer’s disease in women, but not men 51 . The inconsistency in these findings may be due to measurement error because few of the included studies used validated scales measuring loneliness; cultural variation in conceptualization and expression of loneliness; duration and perceived intensity of loneliness exposure; and differences in studies’ adjustment for potentially explanatory covariates especially social isolation, socioeconomic status and depression.
These findings suggest that loneliness but not perceived social support is associated with higher dementia risk, but it is unclear whether this association is only a consequence of having more social contact with others.
Social participation and cognitive decline
Our Review focused on dementia as the most clinically important syndrome, but some studies considered the link between social participation and conditions along the disease pathway such as mild cognitive impairment 52 , providing further supportive evidence. Several studies have also examined the association of social participation with decline in cognitive function, which is relevant as cognitive decline is the core feature of dementia. While associations between potential risk factors and cognitive decline cannot directly indicate elevated dementia risk, the finding of an association is consistent with this possibility 53 . Two systematic reviews of social relationships and cognitive decline found that both the quantity and quality of social relationship participation were linked to subsequent increased risk of cognitive impairment 54 , 55 . However, the methodological quality of included studies precluded strong conclusions about beneficial aspects of social relationships for cognition, and the direction underpinning the association was unclear as 15 of the 19 studies had less than 5 years of follow-up, meaning that reverse causation was likely.
A further review found associations between social activity, networks and support and global cognitive function, although similar limitations apply to this Review 56 . Subsequent studies of German adults over 85 years found that small and declining social networks were associated with worse cognitive decline 57 and this finding was replicated in a study of Japanese adults who were followed for over 10 years 58 . Employment is frequently a source of social contact for many, and previous studies have found that retirement is associated with cognitive decline 59 , 60 , 61 , which may indicate that withdrawal from work-related social networks has detrimental effects on cognitive function. However, leaving employment does not only cause loss of social contact, but may reflect loss of cognitively stimulating occupation, physical labor, social status and income, and the direction of the relationship is not clear as individuals with already declining cognitive function may be more likely to retire if given a choice. The report that countries in which people retire younger have a higher rate of declining cognition compared to countries with later retirement age suggests that there may be some causal relationship 62 . In line with this is the finding of more memory loss in retirees in a study adjusting for health as well as sociodemographic factors 63 .
A systematic review of studies on loneliness and cognitive decline found that loneliness was associated with worse general cognitive function, intelligence quotient, processing speed and delayed and immediate recall 64 . However, confounding factors such as depression and social isolation may also have contributed to these findings. Furthermore, the directional relationship between cognitive impairment and loneliness was not clear as, for example, one study found that poor verbal ability predicted loneliness later in life 65 .
More social network contact and less loneliness is associated with lower cognitive decline, which is consistent with a protective effect against dementia, although methodological limitations in this literature persist. Retirement is associated with cognitive decline, which may partly be due to less social participation.
Limitations of observational evidence
The epidemiological evidence is limited by the probability of reverse causation in studies with a short follow-up that have typically found strong associations of social participation with lower dementia risk. Studies with a longer follow-up have found mixed results and, where a protective effect was reported, the magnitude of associations has generally been small. Whether this is because of lower bias from reverse causation in these studies or greater effect dilution bias due to exposure misclassification remains unclear. In long-term follow-up studies, a single baseline measurement may not accurately indicate social participation across the life course, and the longer the follow-up is, the more likely changes in social participation are to occur, contributing to exposure misclassification, resulting in artificial attenuation of effect estimates. These findings highlight the importance of studies with longer-term follow-up and repeated assessment of social participation, ideally comprising age ranges from early adulthood or even earlier. Such studies would allow further exploration of the life-course influence of social participation on dementia risk, including the ability to identify whether there are critical or sensitive risk periods, or whether an effect of social participation on dementia results from accumulation or chains of risk, guiding future interventions. Ultimately, it is likely that there is a bidirectional relationship between social participation and dementia, whereby low participation accelerates cognitive decline, and this decline impairs social participation.
Confounding is a major source of bias in observational evidence. Associations persisted in studies that adjusted for a wide range of potential confounders, but it is possible that residual confounding persists. Other study designs, such as Mendelian randomization, may help to tackle reverse causation and minimize other sources of confounding, and evidence from one study suggests only partial benefit of social engagement 43 , 66 . There is evidence of publication bias in the literature as reported in two reviews 38 , 54 . Sharing data to combine cohorts in larger, individual participant-data meta-analyses 67 may help to address publication bias, and increase power and generalizability of the findings. Many studies tend to recruit people with lower risk 68 , 69 who are less likely to develop dementia than the general population meaning that findings may not be generalizable.
Most meta-analyses dichotomize the social exposure variables although social participation is not a binary concept, so nuanced examination to clarify dose–response associations and different dimensions of social participation, as used in some studies 41 , would be beneficial. The heterogeneity in social participation measurement related to the different construct definitions and the lack of validated established instruments is another contributing factor to the heterogeneity in findings. There is also limited evidence for whether social participation has a differential effect on different subtypes of dementia, as relatively few studies have examined dementia subtypes. One study found that the association of leisure activity participation was similar for Alzheimer’s dementia and vascular dementia 70 , and results were inconsistent for whether marital status association with dementia differed for Alzheimer’s and vascular etiology 39 .
Social participation is culturally patterned, making it important to understand how the contribution of social participation varies in different settings. Most studies have been conducted in North America, Europe and China, and there is little consistency in the findings (Fig. 2 ). This may be due to differences in study design and measurement of social participation in these studies. It may also reflect that social participation effects differ across settings as the nature of the participation and associated health behaviors vary. The prevalence of social behaviors is also likely to be conditioned by contextual factors, for example, social isolation is reported less commonly in Latin American countries, India and China than the global average 15 . Within countries, there is also ethnic and socioeconomic variation in social participation, for example, in Aotearoa/New Zealand, the contribution of social isolation to dementia risk was greater in European and Māori populations groups than Asian and Pasifika populations 71 , and in Brazil it was greater in poor regions than wealthy ones, although there was no ethnic variation 16 . Interventions therefore need to be designed with consideration to culture and social environments and target different components of social participation to be acceptable and effective in different cultures.
Summary of the findings of studies included in six systematic reviews of observational studies related to social participation and dementia 27 , 37 , 38 , 39 , 44 , 144 , according to the country studied. Studies were divided into those examining social activity engagement, social network contact and support and subjective aspects of social participation. The number of studies of each kind conducted in each country and the consistency of findings are shown. Green indicates countries where all included studies found that more social participation was associated with lower dementia risk (positive association). Red indicates countries where all studies found no association. Orange indicates mixed findings.
In summary, major methodological limitations of previous observational literature on social participation and dementia risk include reverse causation bias, confounding, publication bias, poor generalizability and measurement error. There are few studies in lower-income and middle-income countries, thus we do not know whether these findings generalize to these settings.
Potential underlying mechanisms
Social participation could plausibly result in reduced dementia risk, irrespective of genetic risk 42 , 72 , through one or more mechanisms. These include building cognitive reserve 73 , and better brain maintenance 74 by reducing neurodegeneration by lower vascular damage, reduced stress 75 , promotion of healthy lifestyles and behaviors and enhanced mental stimulation 76 (Fig. 3 ).
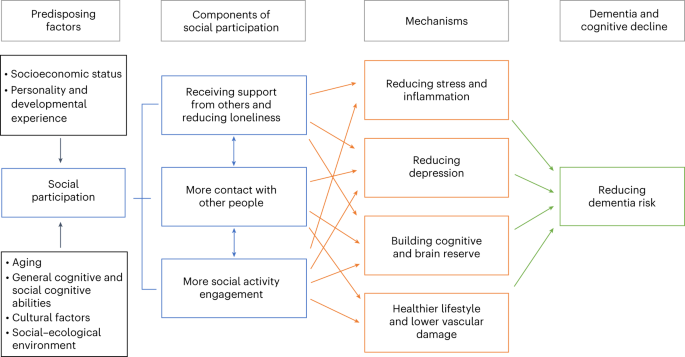
Summary of the mechanisms by which social participation may reduce dementia risk. Neuropsychological factors such as personality, cognition and developmental experiences, and social factors like socioeconomic status and the social and ecological environment predispose individuals to having more or less need for and access to social participation in a range of different domains. Social participation may act through several mechanisms, including brain maintenance by reducing stress, inflammation and cerebrovascular damage, building cognitive reserve and reducing depression to reduce dementia risk.
Cognitive reserve
Social contact with others may build cognitive reserve, implying that increased cognitive adaptability improves the ability of individuals to tolerate neuropathology without corresponding decline in cognition and function 73 . Socialization is cognitively demanding, requiring deployment of numerous social cognitive domains 77 , as well as planning, memory and language and, according to the social brain hypothesis, social complexity may be the major driving force for evolutionary brain development 78 . Social participation may therefore exercise cognitive domains, reducing vulnerability to late-life decline 79 . An autopsy study found that more social contact before death modified the relationship between neuropathology and cognition, such that more amyloid load and neurofibrillary tangle density was less strongly associated with cognitive decline in people who had more frequent social contacts 80 , supporting the hypothesis that cognitive reserve was greater in those with more frequent social contact. Another study found that the detrimental effect of diabetes on dementia risk was attenuated by social participation 81 .
Brain maintenance
Social participation may affect dementia risk through brain maintenance, which refers to reduced accumulation of brain pathology and preservation of a healthy brain conferring better late-life cognitive function 74 . Better social health, defined as less loneliness, more social support and being married, was associated in one study with greater brain volume 82 . Another study showed that socially isolated individuals had lower frontal and temporal gray matter volumes and that differences in gray matter volume partly mediated the association between social isolation at baseline and cognitive function at follow-up 43 . This may partly reflect reverse causation whereby people with larger brains are able to maintain more complex social lives. However, mental stimulation within the occupational context has been linked to greater hippocampal volume 83 and lower levels of proteins that inhibit axonogenesis and synaptogenesis 84 , although it is unclear to what extent the level of mental stimulation during social participation is important. Brain maintenance from social participation may therefore be achieved through several mechanisms.
Lifestyle behaviors and vascular risk
Social participation may improve brain maintenance through healthy lifestyle behaviors. Social isolation is associated with increased mortality through health behaviors such as smoking 85 , high alcohol consumption 86 and cardiovascular diseases 87 . All-cause dementia and dementia subtypes are related to cardiovascular and cerebrovascular health 88 , 89 and heavy alcohol consumption is an established dementia risk factor 14 , so the mechanisms by which lower social contact is associated with cardiovascular risk factors and illness may also extend to dementia risk. Contact with others may, in many cases, model and encourage better health behaviors leading to lower risk of vascular disease and subsequent better cognitive health and lower dementia risk. This is supported by a study of Ecuadorean adults in whom poor social relationships and lower support networks were associated with worse progression of cerebrovascular disease 90 . However, smoking and alcohol consumption may accompany social participation to mitigate socioeconomic stress, so some social contact may not confer cerebrovascular health benefits.
Stress, accelerated aging and inflammation
The effect of social participation on brain maintenance could act through the stress response 91 . A detrimental effect of stress through, for example, restraint, social isolation or other chronic stressors, on hippocampal networks has been demonstrated in animal models 92 , and persistent midlife stress has been associated with elevated dementia risk in human epidemiological studies 93 . Social isolation and poor social support are associated with biological stress markers including dysregulation of the hypothalamic–pituitary–adrenal axis and cortisol responses 94 , increased sympathetic nervous system activation 95 , accelerated cellular aging 96 , 97 , 98 and raised inflammatory markers 99 , suggesting that social isolation may affect dementia risk through the pathological effect of stress. The important role of systemic inflammation in the etiology of dementia is increasingly being recognized 100 , 101 , 102 , 103 .
Some large-scale studies have linked severe infections to increased dementia risk 104 , 105 , suggesting that human-to-human contact could increase dementia risk through the adverse effect of infection. The reduced human contact of socially isolated individuals might therefore potentially decrease their exposure to transmitted pathogens. However, some studies have observed increased risk of severe infections among socially isolated individuals 106 , reflecting overall adverse impacts of social isolation on general health, and this may also reflect the immune suppression linked to stress induced by social isolation 107 . In an analysis making a distinction between subjective loneliness and objective social isolation, the occurrence of hospital-treated infections was higher among lonely people than others, while no robust association was observed between social isolation and the incidence of hospital-treated infections 108 . This preliminary evidence requires further study, and research on long-term impacts of COVID-19-related isolation may be informative.
Social participation, depression and cognitive function are interrelated in a complex fashion. Late-life depression is consistently associated with dementia risk 109 , although this may be partially due to low mood arising in preclinical dementia 110 . However, social participation may arise from and result in low mood, so it is plausible that social participation affects cognition and dementia through affecting mood.
Interventions for social participation
Clarifying the potential of social participation as a target for intervention is crucial but the design of research to test whether increasing social participation affects dementia risk is challenging. It would be unethical to deprive some people of aspects of social participation and impractical to enforce greater social participation in others. While desire for social participation is an inherent aspect of human nature that may have neurobiological underpinnings 111 , and social contact with other people is a core part of individual identity and important for wider society 112 , it is not clear how to improve social participation. Furthermore, some individuals may not wish to change their social participatory habits as, although social contact with others is usually associated with positive affective experiences, personality characteristics such as introversion 113 , autistic traits 114 or internalized stigma related to mental illness 115 may encourage some to seek and prefer solitude.
Local cultures and traditions are important in the roles of individuals within wider society, and an intervention to encourage social participation would need to be acceptable to participants, feasible to administer and sustainable to potentially have an effect. Furthermore, considering the gradual onset of dementia pathology and symptoms, and the likely lifelong contribution of symptoms, interventions may best be delivered at a relatively young age, and many decades of follow-up would be required to assess dementia risk, which is impractical. Interventions have therefore often focused on cognitive change, rather than dementia incidence, as a primary outcome. Randomized controlled trials (RCTs) of social interventions or multicomponent interventions that include a social focus are summarized in Table 2 .
Social interventions for cognitive function
Evidence from studies testing social interventions for people at risk of developing dementia is variable. Interventions for improving cognition through social activities, networks and relationships were summarized in a 2017 systematic review 56 , which found three RCTs, the results of which were not suitable for meta-analysis due to study design heterogeneity. The trials of between 120 and 235 participants tested social activity interventions, which all included facilitator-led group activities. One Finnish study led to a significant but small improvement in cognitive performance on the Alzheimer’s Disease Assessment Scale–Cognitive Subscale (ADAS-cog) 116 at 3 months (1 point difference, −2.6 (3.4, −1.8) versus −1.6 (−2.2, −1.0)) and better subjective cognitive function 117 . The other studies were from China 118 and the USA 119 and showed better but non-significant overall cognitive performance compared to control groups, although one reported improved immediate recall and verbal fluency 118 and a 0.6% increase in total brain volume compared to the control group 118 . These studies were too small and the follow-up time too short to identify any effect on the risk of incident mild cognitive impairment or dementia.
Interventions aimed at improving loneliness in older adults have usually focused on encouraging study participants to engage in purposeful activity and maintain contact with their social networks 120 . Although there is some evidence of efficacy of these programs in reducing loneliness compared to control groups, no studies have assessed effects on cognition 121 .
Interventions to increase social participation through facilitator-led group activities have not consistently been shown in RCTs to improve general cognitive function; studies were too small and short to identify any effect on the development of dementia.
Interventions combining social participation with other strategies
Interventions using a combination of strategies to improve cognition have gained increasing interest as they target several risk factors and can be tailored according to individual risk profiles. A Cochrane review of multi-domain interventions 122 was inconclusive about whether these overall affected dementia risk, but the review determined that only two of these interventions targeted social participation—the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER) RCT 123 and an RCT of Korean community-dwelling adults 124 .
The FINGER study was a 2-year intervention for people with elevated vascular risk for dementia where the intervention consisted of advice on diet, physical activity, cognitive training and improved management of vascular risk. There was no explicit social element to the study, but it was achieved implicitly through group meetings 123 . The intervention resulted in less cognitive decline than the control group with a small effect size (Cohen’s d = 0.13), and this difference persisted irrespective of baseline risk factors 125 , but results using dementia as an outcome have not yet been published. An RCT of a multi-domain intervention delivered over 18 months in Korea included promotion of cognitive and social activities alongside physical activity, alcohol and smoking interventions and weight loss, lean body mass and healthy diet with four different modes of delivery including, at its most intensive, bimonthly health worker-initiated visits, counseling, and rewards for adherence to the program 124 . Only the most intensive approach to delivering the intervention led to better cognitive function compared to the control group at 18 months follow-up on the mini-mental state examination (MMSE; mean performance, 0.99 points better than control (standard error, 0.49)). Both interventions were time-consuming, intensive and costly with small effect sizes for cognition.
A subsequent RCT, SUPERBRAIN, tested a multi-domain intervention including social activities through group meetings and additional scheduled monthly social activities 126 . The Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) score improved significantly at 24 weeks of follow-up of participants in the intervention group who received the intervention in a clinical setting (5.46 ± 7.50, P = 0.004) or at home (5.50 ± 8.14, P = 0.004) compared to the control group (−0.74 ± 11.51) 127 . This improvement was associated with changes in learning and cognitive neural networks 128 .
In all these studies, the specific social contribution to any positive effect is unclear due to the multi-domain nature of the intervention. One study described which health behaviors changed and found that social participation did not significantly increase after the intervention, and analysis instead attributed the cognitive benefit to increased cognitive activity 124 . Other studies to test combination strategies for dementia prevention with interventions, including AgeWell.de 129 , MIND China 130 and US Pointer 131 , are still ongoing.
Interventions targeting several domains including social participation have shown cognitive benefit, although they are time-consuming and costly, and no effect on dementia risk has yet been demonstrated. The specific contribution of social participation to improving cognition is not yet established.
Quasi-experimental research
Considering the challenges of RCTs in this area, quasi-experimental research may be informative. COVID-19-related social isolation measures, introduced to contain the disease, resulted in a reduction in social participation, which affected some, such as those living alone, more than others. The evidence on how this affected dementia risk may become evident in future years. However, some studies have found that social restrictions may have affected cognitive function.
A review of the effect of COVID-19-related isolation on the cognition of people with dementia found that 60% of 15 studies reported a detrimental effect on cognition 23 . Similar findings have been reported in a study of adults without dementia in which adherence to stricter isolation was associated with worse short-term performance on cognitive tasks 132 , but this deterioration of performance may be related to anxiety or affective symptoms. A study found that the link between COVID-19-related social restrictions and poor cognitive performance was more pronounced in younger than older individuals, which would not be expected if poor cognitive performance were related to acceleration, or greater susceptibility to the effects, of neurodegeneration 133 . Findings should be interpreted with caution as reverse causation may partly account for findings; for example, one cross-sectional study of Chinese adults found that worse cognition was associated with lower adherence to COVID-19 prevention regulations 134 and COVID-19 itself may increase risk of dementia 135 . Thus, while isolation measures may be required during periods of high COVID-19 rates or other future infectious disease epidemics, the principle of ‘isolating the virus rather than isolating people’ should be used to decide on measures.
Uncertainties of interventional evidence
There are several uncertainties from the interventional research. Although there is preliminary evidence that group social interventions can improve cognition, there is no evidence about prevention of dementia, and interventions that have demonstrated effect on cognition may not carry through to delay or prevent dementia. However, accelerated cognitive decline begins years before dementia diagnosis and this decline predicts dementia risk 136 . Therefore, the US Food and Drug Administration have encouraged the use of cognitive and functional endpoints, rather than dementia, for drug trials 137 , so improvement in cognition can be considered necessary but not sufficient evidence for identifying dementia prevention strategies. Extended follow-up of previous studies by attaining dementia status from clinical records will support existing evidence.
Additional follow-up may also clarify whether interventions lead to long-lasting changes in social behavior, which would guide timing of an intervention. Considering that dementia results from accumulated lifetime risk, it may be that changing behavior earlier would confer more benefit. The move toward multi-domain interventions, rather than interventions targeting a single risk factor, is reasonable as it allows tailored interventions to change risk profiles. However, it obscures the understanding whether a single component of the intervention is an active ingredient. This may be helped by process evaluation of trials, examination of mechanisms through assessment of biomarkers and analyses that determine adherence to different intervention components and estimate their effects 124 .
Policy implications
Public health policy could promote social participation through several approaches, focusing particularly on the areas with the strongest evidence for dementia prevention, related to social contact and activity engagement (Fig. 4 ). Policy should aim to restore pre-pandemic social participation, including promotion of face-to-face contact and support alongside remote approaches where necessary during periods of high contagion. There should be guidance and support for retirees, because retirement is a potential sensitive period, such as by providing volunteering opportunities and further education to promote social participation. Provision of adequate socially connected housing for older people is a focus of several governmental and third-sector organizations 138 , and has potential to reduce social isolation and loneliness as well as provide support networks for older people. Physical environmental adaptations by optimizing urban planning, accessibility and infrastructure, as recommended in the WHO’s Global Age Friendly Cities : A Guide 139 , could help to promote social inclusion. Finally, provision of leisure time with social connection through social centers would promote social and cognitive activity and participation.
Summary of the potential policy approaches to increasing social participation.
This Review highlights the current evidence for the effect of social participation on dementia risk. There is a pressing need to clarify risk factors for dementia to guide prevention approaches to mitigate the projected future rise in the number of people with dementia. The evidence for a direct causal link between social participation and dementia is not definitive due to limitations in observational research but is consistent and biologically plausible, with evidence for several potential mechanistic pathways underlying an association. There are few RCTs of social interventions and they are consistent with showing a positive effect on cognition, but none include dementia as an outcome.
Evidence has accumulated since the WHO 19 published guidelines on risk reduction for cognitive decline and dementia 19 , which rated evidence on a range of risk factors based on confidence about whether the desirable effects of the intervention outweigh any undesirable effects. The guidelines reported that there was insufficient evidence to recommend social activity for reducing cognitive decline or dementia due to limited and inconclusive evidence and risk of bias arising from reverse causality. There is now considerably more evidence for the link between social participation and dementia, particularly for social contact with others, although the evidence for activity engagement is weaker, suggesting that these guidelines should be reassessed and that future guidelines should highlight social participation as a modifiable risk factor for dementia. There is good evidence that social participation overall is beneficial for general health and well-being 140 and the WHO 141 position paper on optimizing brain health concludes that social connection is an important determinant for brain health across the life course 141 .
Implications for treatment
The US National Academies of Sciences recommend applying quality of evidence standards, which take into account the priority of the problem, how substantial the benefits and harms are, the certainty of the evidence, the value of the outcomes to stakeholders, how an intervention’s clinical and cost effectiveness compares with others, resource requirements, the impact on health equity, the intervention’s acceptability and the feasibility of implementation 142 . These standards could be applied to evaluate social participation interventions. While trials today suggest potential individual-level strategies, there is currently insufficient RCT effectiveness or cost-effectiveness evidence for definitive recommendations, so further research is required on effective and scalable interventions.
Social isolation is closely linked with socioeconomic deprivation 143 , so targeting individuals to improve social participation may be inequitable and ineffective, but health status can be shaped by policies with a societal rather than individual focus. The lower numbers of people with dementia due to higher education standards or reduced smoking are a consequence of societal changes, by, for example, increasing the number of mandatory years in education, and improving public understanding of the harm of, and limiting the opportunities for, cigarette smoking. The key area for applying the understanding that social participation is beneficial for brain health is therefore likely to be in social policies, community awareness, and advice around individuals’ choices related to social participation over the life cycle. These policy approaches should particularly aim to support those from socioeconomic and cultural groups at highest risk of dementia who may find it hardest to access social participation, by reducing financial and logistical barriers to social activities and participation to have a substantial benefit for health and society.
Patterson, C. World Alzheimer report 2018 (Alzheimer’s Disease International, 2018).
Alzheimer’s Research UK. Dementia Attitudes Monitor: Shaping Breakthroughs in Public Understanding of Dementia and Attitudes to Research (2021).
Velandia, P. P. Global and regional spending on dementia care from 2000–2019 and expected future health spending scenarios from 2020–2050: an economic modelling exercise. EClinicalMedicine 45 , 101337 (2022).
Article PubMed PubMed Central Google Scholar
Seshadri, S. & Wolf, P. A. Lifetime risk of stroke and dementia: current concepts, and estimates from the Framingham Study. Lancet Neurol. 6 , 1106–1114 (2007).
Article PubMed Google Scholar
Zissimopoulos, J., Crimmins, E. & Clair, P. S. The value of delaying Alzheimer’s disease onset. Forum Health Econ. Policy 18 , 25–39 (2014).
Röhr, S., Pabst, A., Luck, T. & Riedel-Heller, S. G. Is dementia incidence declining in high-income countries? A systematic review and meta-analysis. Clin. Epidemiol. 10 , 1233–1247 (2018).
Google Scholar
Langa, K. M. et al. A comparison of the prevalence of dementia in the United States in 2000 and 2012. JAMA Intern. Med. 177 , 51–58 (2016).
Article Google Scholar
Matthews, F. et al. A two decade dementia incidence comparison from the Cognitive Function and Ageing Studies I and II. Nat. Commun. 7 , 11398 (2016).
Article CAS PubMed PubMed Central Google Scholar
Wu, Y.-T. et al. The changing prevalence and incidence of dementia over time—current evidence. Nat. Rev. Neurol. 13 , 327–339 (2017).
Sreeramareddy, C. T. & Aye, S. N. Changes in adult smoking behaviours in ten global adult tobacco survey (GATS) countries during 2008–2018—a test of ‘hardening’hypothesis’. BMC Public Health 21 , 1209 (2021).
Mosterd, A. et al. Trends in the prevalence of hypertension, antihypertensive therapy, and left ventricular hypertrophy from 1950 to 1989. N. Engl. J. Med. 340 , 1221–1227 (1999).
Article CAS PubMed Google Scholar
Fang, M., Wang, D., Coresh, J. & Selvin, E. Trends in diabetes treatment and control in US adults, 1999–2018. N. Engl. J. Med. 384 , 2219–2228 (2021).
Kiyohara, Y. Epidemiology of dementia: the Hisayama study. Nihon Rinsho 72 , 601–606 (2014).
PubMed Google Scholar
Livingston, G. et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet 396 , 413–446 (2020).
Mukadam, N., Sommerlad, A., Huntley, J. & Livingston, G. Population attributable fractions for risk factors for dementia in low-income and middle-income countries: an analysis using cross-sectional survey data. Lancet Glob. Health 7 , e596–e603 (2019).
Suemoto, C. K. et al. Risk factors for dementia in Brazil: differences by region and race. Alzheimers Dement. https://doi.org/10.1002/alz.12820 (2022).
Surkalim, D. L. et al. The prevalence of loneliness across 113 countries: systematic review and meta-analysis. BMJ 376 , e067068 (2022).
Serrat, R., Scharf, T., Villar, F. & Gómez, C. Fifty-five years of research into older people’s civic participation: recent trends, future directions. Gerontologist 60 , e38–e51 (2020).
World Health Organization. Risk Reduction of Cognitive Decline and Dementia: WHO Guidelines (2019).
Ang, S. Life course social connectedness: age-cohort trends in social participation. Adv. Life Course Res. 39 , 13–22 (2019).
Dahlberg, L., Agahi, N. & Lennartsson, C. Lonelier than ever? Loneliness of older people over two decades. Arch. Gerontol. Geriatrics 75 , 96–103 (2018).
Sommerlad, A. et al. Social relationships and depression during the COVID-19 lockdown: longitudinal analysis of the COVID-19 Social Study. Psychol. Med. 52 , 3381–3390 (2022).
Suárez-González, A., Rajagopalan, J., Livingston, G. & Alladi, S. The effect of COVID-19 isolation measures on the cognition and mental health of people living with dementia: a rapid systematic review of one year of quantitative evidence. EClinicalMedicine 39 , 101047 (2021).
Levasseur, M. et al. Scoping study of definitions of social participation: update and co-construction of an interdisciplinary consensual definition. Age Ageing 51 , afab215 (2022).
Berkman, L. F. & Syme, S. L. Social networks, host resistance, and mortality: a nine-year follow-up study of Alameda County residents. Am. J. Epidemiol. 109 , 186–204 (1979).
Russell, D., Peplau, L. A. & Cutrona, C. E. The revised UCLA Loneliness Scale: concurrent and discriminant validity evidence. J. Pers. Soc. Psychol. 39 , 472–480 (1980).
Yates, L. A., Ziser, S., Spector, A. & Orrell, M. Cognitive leisure activities and future risk of cognitive impairment and dementia: systematic review and meta-analysis. Int. Psychogeriatr. 28 , 1791–1806 (2016).
Zhou, Z., Wang, P. & Fang, Y. Social engagement and its change are associated with dementia risk among Chinese older adults: a longitudinal study. Sci. Rep. 8 , 1551 (2018).
Fancourt, D., Steptoe, A. & Cadar, D. Community engagement and dementia risk: time-to-event analyses from a national cohort study. J. Epidemiol. Community Health 74 , 71–77 (2020).
Duffner, L. et al. The role of cognitive and social leisure activities in dementia risk: assessing longitudinal associations of modifiable and on-modifiable risk factors. Epidemiol. Psychiatr. Sci. 31 , e5 (2022).
Amieva, H. et al. Prodromal Alzheimer’s disease: successive emergence of the clinical symptoms. Ann. Neurol. 64 , 492–498 (2008).
Jack, C. R. Jr. et al. Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol. 9 , 119–128 (2010).
Almeida-Meza, P., Steptoe, A. & Cadar, D. Is engagement in intellectual and social leisure activities protective against dementia risk? Evidence from the English longitudinal study of ageing. J. Alzheimer’s Dis. 80 , 555–565 (2021).
Nemoto, Y. et al. An additive effect of leading role in the organization between social participation and dementia onset among Japanese older adults: the AGES cohort study. BMC Geriatr. 17 , 297 (2017).
Sommerlad, A. et al. Leisure activity participation and risk of dementia: an 18-year follow-up of the Whitehall II Study. Neurology 95 , e2803–e2815 (2020).
Floud, S. et al. Cognitive and social activities and long-term dementia risk: the prospective UK Million Women Study. Lancet Public Health 6 , e116–e123 (2021).
Kuiper, J. S. et al. Social relationships and risk of dementia: a systematic review and meta-analysis of longitudinal cohort studies. Ageing Res. Rev. 22 , 39–57 (2015).
Penninkilampi, R., Casey, A.-N., Singh, M. F. & Brodaty, H. The association between social engagement, loneliness, and risk of dementia: a systematic review and meta-analysis. J. Alzheimer’s Dis. 66 , 1619–1633 (2018).
Sommerlad, A., Ruegger, J., Singh-Manoux, A., Lewis, G. & Livingston, G. Marriage and risk of dementia: systematic review and meta-analysis of observational studies. J. Neurol. Neurosurg. Psychiatry 89 , 231–238 (2018).
Sundström, A., Westerlund, O., Mousavi-Nasab, H., Adolfsson, R. & Nilsson, L.-G. The relationship between marital and parental status and the risk of dementia. Int. Psychogeriatr. 26 , 749–757 (2014).
Sommerlad, A., Sabia, S., Singh-Manoux, A., Lewis, G. & Livingston, G. Association of social contact with dementia and cognition: 28-year follow-up of the Whitehall II cohort study. PLoS Med. 16 , e1002862 (2019).
Elovainio, M. et al. Association of social isolation, loneliness and genetic risk with incidence of dementia: UK Biobank Cohort Study. BMJ Open 12 , e053936 (2022).
Shen, C. et al. Associations of social isolation and loneliness with later dementia. Neurology 99 , e164–e175 (2022).
Lara, E. et al. Does loneliness contribute to mild cognitive impairment and dementia? A systematic review and meta-analysis of longitudinal studies. Ageing Res. Rev. 52 , 7–16 (2019).
Akhter‐Khan, S. C. et al. Associations of loneliness with risk of Alzheimer’s disease dementia in the Framingham Heart Study. Alzheimer’s Dement. 17 , 1619–1627 (2021).
Sutin, A. R., Stephan, Y., Luchetti, M. & Terracciano, A. Loneliness and risk of dementia. J. Gerontol. B Psychol. Sci. Soc. Sci. 75 , 1414–1422 (2020).
Salinas, J. et al. Association of loneliness with 10-year dementia risk and early markers of vulnerability for neurocognitive decline. Neurology 98 , e1337–e1348 (2022).
Freak-Poli, R. et al. Loneliness, not social support, is associated with cognitive decline and dementia across two longitudinal population-based cohorts. J. Alzheimer’s Dis. 85 , 295–308 (2022).
Shibata, M. et al. Emotional loneliness is associated with a risk of dementia in a general Japanese older population: the Hisayama study. J. Gerontol. B Psychol. Sci. Soc. Sci. 76 , 1756–1766 (2021).
Akhter-Khan, S. et al. Associations of loneliness with risk of Alzheimer’s disease dementia in the Framingham Heart Study. Alzheimers Dement. 17 , 1619–1627 (2021).
Oh, D. J. et al. Association of low emotional and tangible support with risk of dementia among adults 60 years and older in South Korea. JAMA Netw. Open 5 , e2226260–e2226260 (2022).
Gardener, H. et al. Social connectivity is related to mild cognitive impairment and dementia. J. Alzheimer’s Dis. 84 , 1811–1820 (2021).
Weuve, J. et al. Guidelines for reporting methodological challenges and evaluating potential bias in dementia research. Alzheimer’s Dement. 11 , 1098–1109 (2015).
Kuiper, J. S. et al. Social relationships and cognitive decline: a systematic review and meta-analysis of longitudinal cohort studies. Int. J. Epidemiol. 45 , 1169–1206 (2016).
Piolatto, M. et al. The effect of social relationships on cognitive decline in older adults: an updated systematic review and meta-analysis of longitudinal cohort studies. BMC Public Health 22 , 278 (2022).
Kelly, M. E. et al. The impact of social activities, social networks, social support and social relationships on the cognitive functioning of healthy older adults: a systematic review. Syst. Rev. 6 , 259 (2017).
Röhr, S. et al. Changes in social network size are associated with cognitive changes in the oldest-old. Front. Psychiatry 11 , 330 (2020).
Saito, T. et al. Association between intra-individual changes in social network diversity and global cognition in older adults: does closeness to network members make a difference? J. Psychosom. Res. 151 , 110658 (2021).
Roberts, B. A., Fuhrer, R., Marmot, M. & Richards, M. Does retirement influence cognitive performance? The Whitehall II Study. J. Epidemiol. Community Health 65 , 958–963 (2011).
Meng, A., Nexø, M. A. & Borg, V. The impact of retirement on age related cognitive decline—a systematic review. BMC Geriatr. 17 , 160 (2017).
Xue, B. et al. Effect of retirement on cognitive function: the Whitehall II cohort study. Eur. J. Epidemiol. 33 , 989–1001 (2018).
Rohwedder, S. & Willis, R. J. Mental retirement. J. Econ. Perspect. 24 , 119–138 (2010).
Clouston, S. A. & Denier, N. Mental retirement and health selection: analyses from the US Health and Retirement Study. Soc. Sci. Med. 178 , 78–86 (2017).
Boss, L., Kang, D.-H. & Branson, S. Loneliness and cognitive function in the older adult: a systematic review. Int. Psychogeriatr. 27 , 541–553 (2015).
Schnittger, R. I., Wherton, J., Prendergast, D. & Lawlor, B. A. Risk factors and mediating pathways of loneliness and social support in community-dwelling older adults. Aging Ment. Health 16 , 335–346 (2012).
Shen, L.-X. et al. Social isolation, social interaction, and Alzheimer’s disease: a Mendelian randomization study. J. Alzheimer’s Dis. 80 , 665–672 (2021).
Kivimäki, M. et al. IPD-Work consortium: pre-defined meta-analyses of individual-participant data strengthen evidence base for a link between psychosocial factors and health. Scand. J. Work Environ. Health 41 , 312–321 (2015).
Fry, A. et al. Comparison of sociodemographic and health-related characteristics of UK Biobank participants with those of the general population. Am. J. Epidemiol. 186 , 1026–1034 (2017).
Brayne, C. & Moffitt, T. E. The limitations of large-scale volunteer databases to address inequalities and global challenges in health and aging. Nat. Aging 2 , 775–783 (2022).
Verghese, J. et al. Leisure activities and the risk of dementia in the elderly. N. Engl. J. Med. 348 , 2508–2516 (2003).
Ma’u, E., Cullum, S., Cheung, G., Livingston, G. & Mukadam, N. Differences in the potential for dementia prevention between major ethnic groups within one country: a cross-sectional analysis of population attributable fraction of potentially modifiable risk factors in New Zealand. Lancet Reg. Health West. Pac. 13 , 100191 (2021).
Dekhtyar, S. et al. Genetic risk of dementia mitigated by cognitive reserve: a cohort study. Ann. Neurol. 86 , 68–78 (2019).
Stern, Y. et al. Whitepaper: Defining and investigating cognitive reserve, brain reserve, and brain maintenance. Alzheimer’s Dement. 16 , 1305–1311 (2020).
Alvares Pereira, G., Silva Nunes, M. V., Alzola, P. & Contador, I. Cognitive reserve and brain maintenance in aging and dementia: an integrative review. Appl. Neuropsychol. Adult 29 , 1615–1625 (2022).
Grande, G., Qiu, C. & Fratiglioni, L. Prevention of dementia in an ageing world: evidence and biological rationale. Ageing Res. Rev. 64 , 101045 (2020).
Fratiglioni, L., Marseglia, A. & Dekhtyar, S. Ageing without dementia: can stimulating psychosocial and lifestyle experiences make a difference? Lancet Neurol. 19 , 533–543 (2020).
Turkstra, L. S. Conversation-based assessment of social cognition in adults with traumatic brain injury. Brain Inj. 22 , 397–409 (2008).
Lanooij, S. D., Eisel, U. L., Drinkenburg, W. H., van der Zee, E. A. & Kas, M. J. Influencing cognitive performance via social interactions: a novel therapeutic approach for brain disorders based on neuroanatomical mapping? Mol. Psychiatry 28 , 28–33 (2022).
Scarmeas, N. & Stern, Y. Cognitive reserve and lifestyle. J. Clin. Exp. Neuropsychol. 25 , 625–633 (2003).
Bennett, D. A., Schneider, J. A., Tang, Y., Arnold, S. E. & Wilson, R. S. The effect of social networks on the relation between Alzheimer’s disease pathology and level of cognitive function in old people: a longitudinal cohort study. Lancet Neurol. 5 , 406–412 (2006).
Marseglia, A., Wang, H.-X., Rizzuto, D., Fratiglioni, L. & Xu, W. Participating in mental, social, and physical leisure activities and having a rich social network reduce the incidence of diabetes-related dementia in a cohort of Swedish older adults. Diabetes Care 42 , 232–239 (2019).
van der Velpen, I. F. et al. Social health is associated with structural brain changes in older adults: the Rotterdam Study. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 7 , 659–668 (2022).
Maguire, E. A. et al. Navigation-related structural change in the hippocampi of taxi drivers. Proc. Natl Acad. Sci. USA 97 , 4398–4403 (2000).
Kivimäki, M. et al. Cognitive stimulation in the workplace, plasma proteins, and risk of dementia: three analyses of population cohort studies. BMJ 374 , n1804 (2021).
Elovainio, M. et al. Contribution of risk factors to excess mortality in isolated and lonely individuals: an analysis of data from the UK Biobank cohort study. Lancet Public Health 2 , e260–e266 (2017).
Kelly, S., Olanrewaju, O., Cowan, A., Brayne, C. & Lafortune, L. Alcohol and older people: a systematic review of barriers, facilitators and context of drinking in older people and implications for intervention design. PLoS ONE 13 , e0191189 (2018).
Holt-Lunstad, J., Smith, T. B., Baker, M., Harris, T. & Stephenson, D. Loneliness and social isolation as risk factors for mortality: a meta-analytic review. Perspect. Psychol. Sci. 10 , 227–237 (2015).
Larson, E. B. et al. Exercise is associated with reduced risk for incident dementia among persons 65 years of age and older. Ann. Intern. Med. 144 , 73–81 (2006).
Barberger-Gateau, P. et al. Dietary patterns and risk of dementia The Three-City cohort study. Neurology 69 , 1921–1930 (2007).
Del Brutto, O. H., Mera, R. M., Recalde, B. Y., Rumbea, D. A. & Del Brutto, V. J. High social risk influence progression of white matter hyperintensities of presumed vascular origin: a prospective study in community-dwelling older adults. Stroke 53 , 2577–2584 (2022).
Cacioppo, J. T., Cacioppo, S., Capitanio, J. P. & Cole, S. W. The neuroendocrinology of social isolation. Annu. Rev. Psychol. 66 , 733–767 (2015).
Rothman, S. M. & Mattson, M. P. Adverse stress, hippocampal networks, and Alzheimer’s disease. Neuromolecular Med. 12 , 56–70 (2010).
Johansson, L. et al. Common psychosocial stressors in middle-aged women related to longstanding distress and increased risk of Alzheimer’s disease: a 38-year longitudinal population study. BMJ Open 3 , e003142 (2013).
Stafford, M., Gardner, M., Kumari, M., Kuh, D. & Ben-Shlomo, Y. Social isolation and diurnal cortisol patterns in an ageing cohort. Psychoneuroendocrinology 38 , 2737–2745 (2013).
Friedler, B., Crapser, J. & McCullough, L. One is the deadliest number: the detrimental effects of social isolation on cerebrovascular diseases and cognition. Acta Neuropathol. 129 , 493–509 (2015).
Carroll, J. E., Roux, A. V. D., Fitzpatrick, A. L. & Seeman, T. Low social support is associated with shorter leukocyte telomere length in late life: multi-ethnic study of atherosclerosis (MESA). Psychosom. Med. 75 , 171–177 (2013).
Mitchell, A. M., Kowalsky, J. M., Epel, E. S., Lin, J. & Christian, L. M. Childhood adversity, social support, and telomere length among perinatal women. Psychoneuroendocrinology 87 , 43–52 (2018).
Wilson, S. J. et al. Loneliness and telomere length: immune and parasympathetic function in associations with accelerated aging. Ann. Behav. Med. 53 , 541–550 (2019).
Smith, K. J., Gavey, S., RIddell, N. E., Kontari, P. & Victor, C. The association between loneliness, social isolation and inflammation: a systematic review and meta-analysis. Neurosci. Biobehav. Rev. 112 , 519–541 (2020).
Walker, K. A. et al. Systemic inflammation during midlife and cognitive change over 20 years: the ARIC study. Neurology 92 , e1256–e1267 (2019).
CAS PubMed PubMed Central Google Scholar
Sweeney, M. D., Zhao, Z., Montagne, A., Nelson, A. R. & Zlokovic, B. V. Blood–brain barrier: from physiology to disease and back. Physiol. Rev. 99 , 21–78 (2019).
Walker, K. A. et al. Large-scale plasma proteomic analysis identifies proteins and pathways associated with dementia risk. Nat. Aging 1 , 473–489 (2021).
Lindbohm, J. V. et al. Immune system wide biomarker analyses provide causal evidence for autoimmunity in dementia causing diseases. Nat. Aging 2 , 956–972 (2022).
Sipilä, P. N. et al. Hospital-treated infectious diseases and the risk of dementia: a large, multicohort, observational study with a replication cohort. Lancet Infect. Dis. 21 , 1557–1567 (2021).
Sun, J. et al. Hospital-treated infections in early-and mid-life and risk of Alzheimer’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis: a nationwide nested case-control study in Sweden. PLoS Med. 19 , e1004092 (2022).
Bu, F., Philip, K. & Fancourt, D. Social isolation and loneliness as risk factors for hospital admissions for respiratory disease among older adults. Thorax 75 , 597–599 (2020).
Scharf, I. et al. Social isolation causes downregulation of immune and stress response genes and behavioural changes in a social insect. Mol. Ecol. 30 , 2378–2389 (2021).
Elovainio, M. et al. Association of social isolation and loneliness with future risk of hospital-treated infections: a prospective multicohort study. Lancet Public Health 8 , e109–e118 (2023).
Byers, A. L. & Yaffe, K. Depression and risk of developing dementia. Nat. Rev. Neurol. 7 , 323–331 (2011).
Singh-Manoux, A. et al. Trajectories of depressive symptoms before diagnosis of dementia: a 28-year follow-up study. JAMA Psychiatry 74 , 712–718 (2017).
Young, S. N. The neurobiology of human social behaviour: an important but neglected topic. J. Psychiatry Neurosci. 33 , 391–392 (2008).
PubMed PubMed Central Google Scholar
Watts, D. J., Dodds, P. S. & Newman, M. E. Identity and search in social networks. Science 296 , 1302–1305 (2002).
Leary, M. R., Herbst, K. C. & McCrary, F. Finding pleasure in solitary activities: desire for aloneness or disinterest in social contact? Pers. Individ. Dif. 35 , 59–68 (2003).
Chevallier, C., Kohls, G., Troiani, V., Brodkin, E. S. & Schultz, R. T. The social motivation theory of autism. Trends Cogn. Sci. 16 , 231–239 (2012).
Livingston, J. D. & Boyd, J. E. Correlates and consequences of internalized stigma for people living with mental illness: a systematic review and meta-analysis. Soc. Sci. Med. 71 , 2150–2161 (2010).
Rosen, W. G., Mohs, R. C. & Davis, K. L. A new rating scale for Alzheimer’s disease. Am. J. Psychiatry 141 , 1356–1364 (1984).
Pitkala, K. H., Routasalo, P., Kautiainen, H., Sintonen, H. & Tilvis, R. S. Effects of socially stimulating group intervention on lonely, older people’s cognition: a randomized, controlled trial. Am. J. Geriatr. Psychiatry 19 , 654–663 (2011).
Mortimer, J. A. et al. Changes in brain volume and cognition in a randomized trial of exercise and social interaction in a community-based sample of non-demented Chinese elders. J. Alzheimer’s Dis. 30 , 757–766 (2012).
Park, D. C. et al. The impact of sustained engagement on cognitive function in older adults: the Synapse Project. Psychological Sci. 25 , 103–112 (2014).
O’Rourke, H. M., Collins, L. & Sidani, S. Interventions to address social connectedness and loneliness for older adults: a scoping review. BMC Geriatr. 18 , 214 (2018).
Cohen-Mansfield, J. & Perach, R. Interventions for alleviating loneliness among older persons: a critical review. Am. J. Health Promot. 29 , e109–e125 (2015).
Hafdi, M., Hoevenaar-Blom, M. P. & Richard, E. Multi‐domain interventions for the prevention of dementia and cognitive decline. Cochrane Database Syst. Rev. 11 , CD013572 (2021).
Ngandu, T. et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): a randomised controlled trial. Lancet 385 , 2255–2263 (2015).
Lee, K. S. et al. Effects of a multidomain lifestyle modification on cognitive function in older adults: an eighteen-month community-based cluster randomized controlled trial. Psychother. Psychosom. 83 , 270–278 (2014).
Rosenberg, A. et al. Multidomain lifestyle intervention benefits a large elderly population at risk for cognitive decline and dementia regardless of baseline characteristics: the FINGER trial. Alzheimer’s Dement. 14 , 263–270 (2018).
Park, H. K. et al. South Korean study to prevent cognitive impairment and protect brain health through lifestyle intervention in at-risk elderly people: protocol of a multicenter, randomized controlled feasibility trial. J. Clin. Neurol. 16 , 292–303 (2020).
Moon, S. Y. et al. Facility-based and home-based multidomain interventions including cognitive training, exercise, diet, vascular risk management, and motivation for older adults: a randomized controlled feasibility trial. Aging 13 , 15898–15916 (2021).
Moon, S. Y. et al. Impact of a multidomain lifestyle intervention on regional spontaneous brain activity. Front. Aging Neurosci. 14 , 926077 (2022).
Zülke, A. et al. AgeWell.de—study protocol of a pragmatic multi-center cluster-randomized controlled prevention trial against cognitive decline in older primary care patients. BMC Geriatr. 19 , 203 (2019).
Wang, Y. et al. MIND China (China) World‐wide FINGERS network: the first global network of multidomain dementia prevention trials. Alzheimer’s Dement. 16 , e046957 (2020).
Baker, L. D. et al. US POINTER (USA) World‐wide FINGERS network: the first global network of multidomain dementia prevention trials. Alzheimer’s Dement. 16 , e046951 (2020).
Ingram, J., Hand, C. J. & Maciejewski, G. Social isolation during COVID‐19 lockdown impairs cognitive function. Appl. Cogn. Psychol. 35 , 935–947 (2021).
Menze, I., Mueller, P., Mueller, N. G. & Schmicker, M. Age-related cognitive effects of the COVID-19 pandemic restrictions and associated mental health changes in Germans. Sci. Rep. 12 , 8172 (2022).
Shen, F., Min, C., Lu, Y. & Chu, Y. The effect of cognition and affect on preventive behaviors during the COVID-19 pandemic: a cross-sectional study in China. BMC Public Health 21 , 722 (2021).
Xu, E., Xie, Y. & Al-Aly, Z. Long-term neurologic outcomes of COVID-19. Nat. Med. 28 , 2406–2415 (2022).
Karr, J. E., Graham, R. B., Hofer, S. M. & Muniz-Terrera, G. When does cognitive decline begin? A systematic review of change point studies on accelerated decline in cognitive and neurological outcomes preceding mild cognitive impairment, dementia, and death. Psychol. Aging 33 , 195–218 (2018).
Yu, J. C., Hlávka, J. P., Joe, E., Richmond, F. J. & Lakdawalla, D. N. Impact of non‐binding FDA guidances on primary endpoint selection in Alzheimer’s disease trials. Alzheimer’s Dement. 8 , e12280 (2022).
AGE Platform Europe. The Right to Adequate Housing of Older Persons; Age Platform Europe Contribution (2022).
World Health Organization. Global Age-Friendly Cities: A Guide (World Health Organization, 2007).
Holt-Lunstad, J., Smith, T. B. & Layton, J. B. Social relationships and mortality risk: a meta-analytic review. PLoS Med. 7 , e1000316 (2010).
World Health Organization. Optimizing Brain Health Across the Life Course: WHO Position Paper (2022).
National Academies of Sciences, E. & Medicine. Evidence-Based Practice for Public Health Emergency Preparedness and Response (National Academies Press, 2020).
Röhr, S. et al. Social factors and the prevalence of social isolation in a population-based adult cohort. Soc. Psychiatry Psychiatr. Epidemiol 57 , 1959–1968 (2021).
Valenzuela, M. J. & Sachdev, P. Brain reserve and dementia: a systematic review. Psychol. Med. 36 , 441–454 (2006).
Berkman, L. F., Glass, T., Brissette, I. & Seeman, T. E. From social integration to health: Durkheim in the new millennium. Soc. Sci. Med. 51 , 843–857 (2000).
Leedahl, S. N., Sellon, A. & Chapin, R. K. Assessment of multiple constructs of social integration for older adults living in nursing homes. J. Gerontol. Soc. Work 61 , 526–548 (2018).
Zavaleta, D., Samuel, K. & Mills, C. Social Isolation: A Conceptual and Measurement Proposal. OPHI Working Papers 67. Report no. 1907194541 (2014).
Perlman, D. & Peplau, L. A. in Preventing the Harmful Consequences of Severe and Persistent Loneliness vol. 13 (eds. L. A. Peplau & S. Goldston) 13–46 (US Government Printing Office, 1984).
Stringhini, S. et al. Socioeconomic status, structural and functional measures of social support, and mortality: The British Whitehall II Cohort Study, 1985–2009. Am. J. Epidemiol. 175 , 1275–1283 (2012).
Download references
Acknowledgements
This research was funded in whole, or in part, by the Wellcome Trust (222932/Z/21/Z). For the purpose of open access, the author has applied a CC BY public copyright license to any author-accepted manuscript version arising from this submission. A.S. and G.L. are supported by the University College London Hospitals National Institute for Health Research (NIHR) Biomedical Research Centre and funded by the NIHR ENHANCE study (NIHR203670). M.K. was supported by the Wellcome Trust (221854/Z/20/Z), the UK Medical Research Council (MRC; S011676/1, R024227/1), the US National Institute on Aging (R01AG056477) and the Academy of Finland (350426). E.B.L. is supported by research grants from the US National Institute on Aging (UO1AG0006781, RO1AG). S.R. is a Global Atlantic Fellow for Equity in Brain Health and supported by the Global Brain Health Institute. A.S.-M. is supported by grants from the National Institute on Aging, National Institutes of Health (R01AG056477, RF1AG062553). G.L. is supported by NIHR ARC and is an NIHR senior investigator.
Author information
Authors and affiliations.
Division of Psychiatry, University College London, London, UK
Andrew Sommerlad, Mika Kivimäki, Archana Singh-Manoux & Gill Livingston
Camden and Islington NHS Foundation Trust, London, UK
Andrew Sommerlad & Gill Livingston
Department of Public Health, University of Helsinki, Helsinki, Finland
Mika Kivimäki
University of Washington Schools of Medicine and Public Health, Seattle, WA, USA
Eric B. Larson
School of Psychology, Massey University, Manawatu, New Zealand
Susanne Röhr
Global Brain Health Institute (GBHI), Trinity College Dublin, Dublin, Ireland
Institute of Social Medicine, Occupational Health and Public Health (ISAP), Medical Faculty, University of Leipzig, Leipzig, Germany
Department of Social Medicine, Graduate School of Medicine, Osaka University, Osaka, Japan
Kokoro Shirai
Université Paris Cité, Inserm, U1153, Paris, France
Archana Singh-Manoux
You can also search for this author in PubMed Google Scholar
Contributions
A.S. and G.L. conceived the review. A.S. reviewed the literature and drafted the article. M.K., E.B.L., S.R., K.S. and A.S.-M. contributed to the redrafting of the article and approved the final version for submission.
Corresponding author
Correspondence to Andrew Sommerlad .
Ethics declarations
Competing interests.
The authors declare no competing interests.
Peer review
Peer review information.
Nature Aging thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Reprints and permissions
About this article
Cite this article.
Sommerlad, A., Kivimäki, M., Larson, E.B. et al. Social participation and risk of developing dementia. Nat Aging 3 , 532–545 (2023). https://doi.org/10.1038/s43587-023-00387-0
Download citation
Received : 29 September 2022
Accepted : 22 February 2023
Published : 18 May 2023
Issue Date : May 2023
DOI : https://doi.org/10.1038/s43587-023-00387-0
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
This article is cited by
Relationship between social participation and depressive symptoms in patients with multimorbidity: the chained mediating role of cognitive function and activities of daily living.
- Shihong Dong
BMC Public Health (2024)
High cognitive reserve attenuates the risk of dementia associated with cardiometabolic diseases
- Abigail Dove
- Wenzhe Yang
Alzheimer's Research & Therapy (2024)
Childhood adversities and memory function in later life: the mediating role of activity participation
BMC Geriatrics (2024)
Comparison of the Therapeutic Effects of Isokinetic and Isotonic Training in Patients with Mild Cognitive Impairment
- Ting-Hsu Lee
- Chiann-Yi Hsu
- Yuan-Yang Cheng
Journal of Cognitive Enhancement (2024)
Association between adult education, brain volume and dementia risk: longitudinal cohort study of UK Biobank participants
- Andrew Sommerlad
- Naaheed Mukadam
GeroScience (2024)
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.

IMAGES